A) \[NaHC{{O}_{3}}\]
B) \[NaOH\]
C) \[Na\]
D) \[PC{{l}_{3}}\]
Correct Answer: A
Solution :
Phenol and benzoic acid can be distinguished by \[NaHC{{O}_{3}}\] because being stronger acid only benzoic acid reacts with it and phenol does not react. Effervescences of \[C{{O}_{2}}\] confirms presence of \[COOH\] group.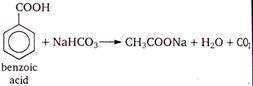
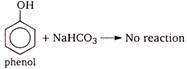 Phenol and benzoic acid react with other reagents \[(NaOH,\,\,Na,\,\,PC{{l}_{5}})\] given as choices. So, they cannot be used to distinguish them.
Phenol and benzoic acid react with other reagents \[(NaOH,\,\,Na,\,\,PC{{l}_{5}})\] given as choices. So, they cannot be used to distinguish them.
You need to login to perform this action.
You will be redirected in
3 sec
