A) beryl
B) zeolite
C) talc
D) feldspar
Correct Answer: A
Solution :
If two oxygen atoms per tetrahedron are shared to form closed rings such that the structures with the general formula \[(SiO_{3}^{2-})\] or \[(Si{{O}_{3}})_{n}^{2n-}\] are obtained, the silicates containing these anions are called cyclic silicates. For example, in beryl \[(B{{e}_{3}}A{{l}_{2}}S{{i}_{6}}{{O}_{18}})\] the anion \[S{{i}_{6}}O_{18}^{12-}\] is cyclic silicate.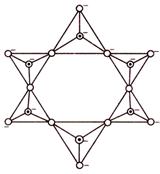
You need to login to perform this action.
You will be redirected in
3 sec
