| (a) Why are most carbon compounds poor conductors of electricity? |
| (b) Write the name and structure of a saturated compound in which the carbon atoms are arranged in a ring. Give the number of single bonds present in this compound. |
Answer:
(a) Electricity is conducted by moving electrons. But carbon forms covalent bonds by sharing of electrons. It does not have free electrons.
(b) Cyclohexane is a saturated compound in which carbon atoms are arranged in a ring.
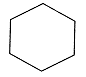
Cyclohexane
There are 6 single bonds present in this compound.
You need to login to perform this action.
You will be redirected in
3 sec
