A) \[ClO_{3}^{-}\]
B) \[ClO_{4}^{-}\]
C) \[NO_{3}^{-}\]
D) \[CO_{3}^{2-}\]
Correct Answer: B
Solution :
The resonating structures of\[ClO_{4}^{-}\]are as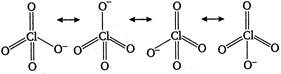 Bond order \[=\frac{total\text{ }number\text{ }of\text{ }bonds\text{ }between\text{ }Cl\text{ }and\text{ O}}{total\text{ }number\text{ }of\text{ }resonating\text{ }structures}\] \[=\frac{7}{4}=1.75\]
Bond order \[=\frac{total\text{ }number\text{ }of\text{ }bonds\text{ }between\text{ }Cl\text{ }and\text{ O}}{total\text{ }number\text{ }of\text{ }resonating\text{ }structures}\] \[=\frac{7}{4}=1.75\]
You need to login to perform this action.
You will be redirected in
3 sec
