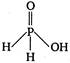
A)
B)
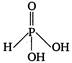
C)
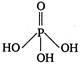
D)
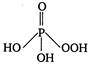
Correct Answer: A
Solution :
Hypophosphorous acid\[({{H}_{3}}P{{O}_{2}})\]has one\[P-OH\]and two\[P-H\]bonds and hence, its structural formula is as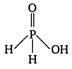
You need to login to perform this action.
You will be redirected in
3 sec
