A) \[n=2\] to \[n=1\] and \[n=3\] to \[n=1\] respectively
B) \[n=2\] to \[n=1\] and \[n=3\] to \[n=2\]respectively
C) \[n=3\] to \[n=2\] and \[n=4\] to \[n=2\]respectively
D) \[n=3\] to \[n=2\] and \[n=4\] to \[n=3\]respectively
Correct Answer: A
Solution :
When the colliding electron remove an electron from innermost K-shell (corresponding to n = 1) of atom and electron from some higher shell jumps to K-shell to fill up this vacancy, characteristic X-ray of K-series are obtained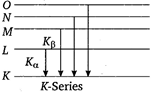 \[\therefore \]\[{{K}_{\alpha }}\] and \[{{K}_{\beta }}\] X-rays are emitted when there is transition of electron between the levels \[n=2\] to \[n=1\]and \[n=3\] to \[n=1\] respectively.
\[\therefore \]\[{{K}_{\alpha }}\] and \[{{K}_{\beta }}\] X-rays are emitted when there is transition of electron between the levels \[n=2\] to \[n=1\]and \[n=3\] to \[n=1\] respectively.
You need to login to perform this action.
You will be redirected in
3 sec
