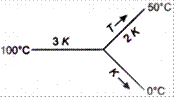
 Then, the temperature of the junction in steady state is
Then, the temperature of the junction in steady state is
A) \[2.0\,m{{s}^{-1}}\]
B) \[2.5\,m{{s}^{-1}}\]
C) \[75{}^\circ C\]
D) \[\frac{50}{3}\] °C
Correct Answer: A
Solution :
According to the figure \[H={{H}_{1}}+{{H}_{2}}\] \[\Rightarrow \] \[\frac{3KA(100-T)}{l}=\frac{2K(T-50)}{l}+\frac{KA(T-0)}{l}\] \[300-3T=2T-100+T\] \[\Rightarrow \] \[6T=400\] or \[T=\frac{200}{3}{{\,}^{o}}C\]You need to login to perform this action.
You will be redirected in
3 sec
