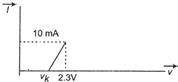
A) 5 \[k\Omega \]
B) 0.2 \[k\Omega \]
C) 2.3 \[k\Omega \]
D) \[\left( \frac{10}{2.3} \right)k\Omega \]
Correct Answer: B
Solution :
\[{{H}_{2}}+\frac{1}{2}{{O}_{2}}\to {{H}_{2}}O;\Delta H=-68.3\] \[{{C}_{2}}{{H}_{6}}+\frac{3}{2}{{O}_{2}}\to 2C{{O}_{2}}+3{{H}_{2}}O\] \[\Delta \text{x}=2\left( -94.1 \right)+3\left( -68.3 \right)-\left( -21.1 \right)\] \[\underset{s}{\mathop{A{{g}_{2}}Cr{{O}_{4}}}}\,\to \underset{2s}{\mathop{2A{{g}^{+}}}}\,+\underset{s}{\mathop{CrO_{4}^{2-}}}\,\] = 0.2 \[{{K}_{sp}}={{\left( 2s \right)}^{2}}s=4{{s}^{3}}\]You need to login to perform this action.
You will be redirected in
3 sec
