A) \[Zn,HCl\]
B) \[LiAl{{H}_{4}}\]
C) \[Mg,\] anhydrous ether,\[{{H}_{2}}O\]
D) \[{{B}_{2}}{{H}_{6}}\] in THF
Correct Answer: D
Solution :
\[Zn+HCl,LiAl{{H}_{4}}\]and Mg, anhydrous ether\[+{{H}_{2}}O\] etc., convert n-butyl chloride into n-butane but \[{{B}_{2}}{{H}_{6}}\]in THF does not convert n-butyl chloride into n-butane. \[{{C}_{4}}{{H}_{9}}Cl\xrightarrow{Zn\,+HCl}\underset{n-\text{butane}}{\mathop{{{C}_{4}}{{H}_{10}}}}\,\] \[{{C}_{4}}{{H}_{9}}Cl+2H\xrightarrow[-2HCl]{LiAl{{H}_{4}}}\underset{n-\text{butane}}{\mathop{{{C}_{4}}{{H}_{10}}}}\,\] \[{{C}_{4}}{{H}_{9}}Cl+Mg\xrightarrow[\text{ether}]{\text{Anhydrous}}{{C}_{4}}{{H}_{9}}MgCl\xrightarrow{{{H}_{2}}O}\]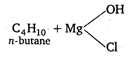
You need to login to perform this action.
You will be redirected in
3 sec
