A) \[Te{{F}_{2}}\]
B) \[ICl_{2}^{-}\]
C) \[SbC{{l}_{3}}\]
D) \[BaC{{l}_{2}}\]
Correct Answer: B
Solution :
Species having the same number of bond pairs and lone pairs are isostructural (have same structure).| Species | Ip + bp | Structure |
| \[Xe{{F}_{2}}\] | \[4lp+2bp\] | Linear 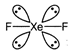 |
| \[Te{{F}_{2}}\] | \[2lp+2bp\] | Angular or V-shape |
| \[ICl_{2}^{-}\] | \[4lp+2bp\] | Linear 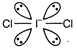 |
| \[BaC{{l}_{2}}\] | \[0lp+2bp\] | \[Cl-Ba-Cl(linear)\] |
You need to login to perform this action.
You will be redirected in
3 sec
