question_answer 1) A weight w is suspended from the midpoint of a rope, whose ends are at the same level. In order to make the rope perfectly horizontal, the force applied to each of its ends must be:
A)
less than w
done
clear
B)
equal to w
done
clear
C)
equal to 2w
done
clear
D)
infinitely large
done
clear
View Answer play_arrow
question_answer 2) A velocity of a particle at an instant is 10 m/s. After 3 s its velocity will become 16 m/s. The velocity at 2 s, before the given instant will be :
A)
6 m/s
done
clear
B)
4 m/s
done
clear
C)
2 m/s
done
clear
D)
1m/s
done
clear
View Answer play_arrow
question_answer 3) A heavy stone hanging from a massless string of length 15 m is projected horizontally with speed 147 m/ s. The speed of the particle at the point where the tension in the string equals the weight of the particle is?
A)
10 m/s
done
clear
B)
7 m/s
done
clear
C)
12 m/s
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 4) When a body moves with constant speed in a circular path, then :
A)
work done will be zero
done
clear
B)
acceleration will be zero
done
clear
C)
no force acts on the body
done
clear
D)
its velocity remains constant
done
clear
View Answer play_arrow
question_answer 5) Two stones are projected with same velocity v at an angle \[\theta \] and \[(90-\,\theta )\] . If \[H\,and\text{ }{{H}_{1}}\] are the greatest heights in the two paths, what is the relation between R, H and \[{{H}_{1}}\]?
A)
\[R=4\frac{\sqrt{H{{H}_{1}}}}{{}}\]
done
clear
B)
\[R=\frac{\sqrt{H{{H}_{1}}}}{{}}\]
done
clear
C)
\[R=4H{{H}_{1}}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 6) A body initially at rest is moving with uniform acceleration a. Its velocity after n s is v. The displacement of the body in 2 s is :
A)
\[\frac{2v(n-1)}{n}\]
done
clear
B)
\[\frac{v(n-1)}{n}\]
done
clear
C)
\[\frac{v(n+1)}{n}\]
done
clear
D)
\[\frac{2v(n+1)}{n}\]
done
clear
View Answer play_arrow
question_answer 7) The magnetic force on a point charge is \[\overrightarrow{\mathbf{F}}\text{=q(}\overrightarrow{\mathbf{V}}\text{ }\!\!\times\!\!\text{ }\overrightarrow{\mathbf{B}}\text{)}\] Here, \[q\] = electric charge \[\overrightarrow{\mathbf{V}}\]= velocity of point charge \[\overrightarrow{\mathbf{B}}\] = magnetic Held The dimension of \[\overrightarrow{\mathbf{B}}\] is :
A)
\[[ML{{T}^{-1}}A]\]
done
clear
B)
\[[{{M}^{2}}L{{T}^{2}}{{A}^{-1}}]\]
done
clear
C)
\[[M{{T}^{-2}}{{A}^{-1}}]\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 8) The first diffraction minimum due to single slit diffraction is \[\theta \], for a light of wavelength \[5000\overset{\text{o}}{\mathop{\text{A}}}\,\]. If the width of the slit is \[1\times {{10}^{-4}},\] then the value of \[\text{ }\!\!\theta\!\!\text{ }\] is :
A)
\[30{}^\circ \]
done
clear
B)
\[45{}^\circ \]
done
clear
C)
\[60{}^\circ \]
done
clear
D)
\[15{}^\circ \]
done
clear
View Answer play_arrow
question_answer 9) A stone of mass m tied to a string of length \[l\]is rotated in a circle with the other end of the string as the centre. The speed of the stone is v. If the string breaks, the stone will:
A)
move towards the centre
done
clear
B)
move away from the centre
done
clear
C)
move along tangent
done
clear
D)
stop
done
clear
View Answer play_arrow
question_answer 10) A_ particle moves with a velocity \[\text{(5}\mathbf{\hat{i}}\text{-3\hat{J}+6\hat{K})}\] m/s under the influence of a constant force \[\overrightarrow{\mathbf{F}}\text{=(10}\mathbf{\hat{i}}\text{+10}\mathbf{\hat{J}}\text{+12}\mathbf{\hat{K}}\text{)}\] instantaneous power applied to the particle is:
A)
200 J/s
done
clear
B)
40J/s
done
clear
C)
140 J/s
done
clear
D)
170 J/s
done
clear
View Answer play_arrow
question_answer 11) The potential energy of a particle of mass 5 kg moving in the \[x-y\] plane is given by \[\text{U=(7x+24y-)J}\text{.x}\,\text{and}\,\text{y}\] being in meter. If the particle starts from rest from origin, then speed of panicle at t = 2 s is:
A)
5m/s
done
clear
B)
14m/
done
clear
C)
17.5m/s
done
clear
D)
10 m/s
done
clear
View Answer play_arrow
question_answer 12) The ratio of radii of gyration of a circular disc and a circular ring of the same radii and same mass about a tangential axis in the plane is :
A)
1; 2
done
clear
B)
\[\sqrt{5}:\sqrt{6}\]
done
clear
C)
2:3
done
clear
D)
2:1
done
clear
View Answer play_arrow
question_answer 13) If a particle of mass m is projected at an angle a with the horizontal, then :
A)
the angular momentum remain constant
done
clear
B)
the linear momentum of particle remains constant
done
clear
C)
total mechanical energy remains constant in the absence of air resistance
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 14) A satellite is moving on a circular path of radius r around the earth has a time period T. If its radius slightly increases by \[\Delta r\], the change in its time period is :
A)
\[\frac{3}{2}\left( \frac{T}{r} \right)\Delta r\]
done
clear
B)
\[\left( \frac{T}{r} \right)\Delta r\]
done
clear
C)
\[\frac{3}{2}\left( \frac{{{T}^{2}}}{{{r}^{2}}} \right)\Delta r\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 15) A particle executes SHM, its time period is 16 s. If it passes through the centre of oscillation the en its velocity is 2m/s at time 2 s. The amplitude will be:
A)
7.2 m
done
clear
B)
4 cm
done
clear
C)
6 cm
done
clear
D)
0.72m
done
clear
View Answer play_arrow
question_answer 16) A simple harmonic oscillator has amplitude A, angular velocity \[\Omega \], and mass m. Then, average energy in one time period will be :
A)
\[\frac{1}{4}m{{\omega }^{2}}{{A}^{2}}\]
done
clear
B)
\[\frac{1}{4}{{m}^{2}}{{\omega }^{2}}{{A}^{2}}\]
done
clear
C)
\[{{\omega }^{2}}{{A}^{2}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 17) A particle executes simple harmonic motion with a frequency\[f\]. The frequency with which the potential energy oscillates is :
A)
\[f\]
done
clear
B)
\[f/2\]
done
clear
C)
\[2f\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 18) The wave front due to a source situated at infinity is :
A)
spherical
done
clear
B)
cylindrical
done
clear
C)
planar
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 19) An astronaut is approaching the moon. He sends out a radio signal of frequency 5000 MHz and the frequency of echo is different from that of the original frequency by 100 kHz. His velocity of approach with respect to the moon is:
A)
2km/s
done
clear
B)
3km/s
done
clear
C)
4km/s
done
clear
D)
5km/s
done
clear
View Answer play_arrow
question_answer 20) The temperature of \[{{H}_{2}}\] at which the rms velocity of its molecules is seven times the rms velocity of the molecules of nitrogen at 300 K is :
A)
2100 K
done
clear
B)
1700 K
done
clear
C)
1350 K
done
clear
D)
1050 K
done
clear
View Answer play_arrow
question_answer 21) Gas exerts pressure on the walls of the container because :
A)
gas has weight
done
clear
B)
gas molecules have momentum
done
clear
C)
gas molecules collide with each other
done
clear
D)
gas molecules collide with the walls of the
done
clear
View Answer play_arrow
question_answer 22) Container The inside and outside temperatures of a refrigerator are 273 K and 303 K respectively. Assuming that refrigerator cycle is reversible, for every joule of work done, the heat delivered to the surrounding will be :
A)
10 J
done
clear
B)
20 J
done
clear
C)
30 J
done
clear
D)
50 J
done
clear
View Answer play_arrow
question_answer 23) In an energy recycling process, X g of steam at 100°C becomes water at 100°C which converts Y g of ice at 0°C into water at 100°C. The ratio of X and Y will be:
A)
\[\frac{1}{3}\]
done
clear
B)
\[\frac{1}{3}\]
done
clear
C)
3
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 24) A body at a temperature of \[728{}^\circ C\] and has surface area \[5\text{ }c{{m}^{2}}\], radiates 300 J of energy each minute. The emissivity is : (Given: Boltzmann constant \[\text{=5}\text{.67 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-8}}}\text{W}{{\text{m}}^{\text{2}}}{{\text{K}}^{\text{4}}}\]
A)
e=0.18
done
clear
B)
e=0.02
done
clear
C)
e=0.2
done
clear
D)
e=0.15
done
clear
View Answer play_arrow
question_answer 25) If \[\sigma =\]surface charge density, \[\varepsilon =\] electric permittivity, the dimensions of\[\frac{\sigma }{\varepsilon }\] are same as:
A)
electric force
done
clear
B)
electric field intensity
done
clear
C)
pressure
done
clear
D)
electric charge
done
clear
View Answer play_arrow
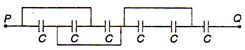
question_answer 26)
For circuit the equivalent capacitance between P and Q is:
A)
6C
done
clear
B)
4C
done
clear
C)
\[\frac{3C}{2}\]
done
clear
D)
\[\frac{6C}{11}\]
done
clear
View Answer play_arrow
question_answer 27) A wire has resistance 120. It is bent in the form of a circle. The effective resistance between the two points on any diameter of the circle is :
A)
12\[\Omega \]
done
clear
B)
24\[\Omega \]
done
clear
C)
6\[\Omega \]
done
clear
D)
3\[\Omega \]
done
clear
View Answer play_arrow
question_answer 28) If two identical heaters each rated as (1000W, 220V) are connected in parallel to 220 V, then the total power consumed is :
A)
200 W
done
clear
B)
2500 W
done
clear
C)
250 W
done
clear
D)
2000 W
done
clear
View Answer play_arrow
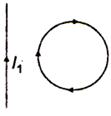
question_answer 29)
In the given figure, the loop is fixed but straight wire can move. The straight wire will:
A)
remain stationary
done
clear
B)
move towards the loop
done
clear
C)
move away from the loop
done
clear
D)
rotates about the axis
done
clear
View Answer play_arrow
question_answer 30) At a point on the right bisector of a magnetic dipole, the magnetic :
A)
potential varies as \[\frac{1}{r2}\]
done
clear
B)
potential is zero at all points on the right bisector
done
clear
C)
field varies as r3
done
clear
D)
field is perpendicular to the axis of the dipole
done
clear
View Answer play_arrow
question_answer 31) The couple acting on a magnet of length 10 cm and pole strength 15 Am, kept in a field of \[B=2~\times {{10}^{-5}}T,\] at an angle of 30° is :
A)
\[1.5\times {{10}^{-5}}Nm\]
done
clear
B)
\[1.5\times {{10}^{-3}}Nm\]
done
clear
C)
\[1.5\times {{10}^{-2}}Nm\]
done
clear
D)
\[1.5\times {{10}^{-6}}Nm\]
done
clear
View Answer play_arrow
question_answer 32) In a step-up transformer, the number of turms in:
A)
primary are less
done
clear
B)
primary are more
done
clear
C)
primary and secondary are equal
done
clear
D)
primary are infinite
done
clear
View Answer play_arrow
question_answer 33) If a circuit made up of a resistance \[\Omega \] and inductance 0.01 H, and alternating emf 200 V at SO Hz is connected, then the phase difference between the current and the emf in the circuit is :
A)
\[{{\tan }^{-1}}(\pi )\]
done
clear
B)
\[{{\tan }^{-1}}\left( \frac{\pi }{2} \right)\]
done
clear
C)
\[{{\tan }^{-1}}\left( \frac{\pi }{4} \right)\]
done
clear
D)
\[{{\tan }^{-1}}\left( \frac{\pi }{3} \right)\]
done
clear
View Answer play_arrow
question_answer 34) An AC is represented by e = 220 sin (100 \[\pi \]) t V and is applied over a resistance of 110 \[\Omega \]. The heat produced in 7 min is :
A)
\[11\times {{10}^{3}}cal\]
done
clear
B)
\[22\times {{10}^{3}}cal\]
done
clear
C)
\[33\times {{10}^{3}}cal\]
done
clear
D)
\[25\times {{10}^{3}}cal\]
done
clear
View Answer play_arrow
question_answer 35) The wavelength of a radio wave of frequency of 1 MHz is :
A)
400m
done
clear
B)
300m
done
clear
C)
350m
done
clear
D)
200m
done
clear
View Answer play_arrow
question_answer 36) The correct option, if speed of \[\gamma \]-rays, x-rays and micro waves are\[{{\gamma }_{g,}}{{v}_{x}}\]and \[{{v}_{m}}\] respectively will be:
A)
\[{{v}_{g}}>{{v}_{g}}>{{v}_{m}}\]
done
clear
B)
\[{{v}_{g}}>{{v}_{x}}>{{v}_{m}}\]
done
clear
C)
\[{{v}_{g}}>{{v}_{x}}>{{v}_{m}}\]
done
clear
D)
\[{{v}_{g}}>{{v}_{x}}>{{v}_{m}}\]
done
clear
View Answer play_arrow
question_answer 37) The electric field E and magnetic field B in electromagnetic waves are:
A)
parallel to each other
done
clear
B)
inclined at an angle of 45°
done
clear
C)
perpendicular to each other
done
clear
D)
opposite to each other
done
clear
View Answer play_arrow
question_answer 38) In Youngs double slit experiment, the spacing between the slits is d and wavelength of light used is 6000 Å. If the angular width of a fringe formed on a distant screen is 1°, then value of d is:
A)
1 mm
done
clear
B)
0.05 mm
done
clear
C)
0.03 mm
done
clear
D)
0.01 mm
done
clear
View Answer play_arrow
question_answer 39) Polarisation of light proves :
A)
corpuscular nature of light
done
clear
B)
quantum nature of light
done
clear
C)
transverse wave nature of light
done
clear
D)
longitudinal wave nature of light
done
clear
View Answer play_arrow
question_answer 40) In terms of Rydberg constant R, the wave number of the first Balmer line is :
A)
R
done
clear
B)
3R
done
clear
C)
\[\frac{\text{5R}}{\text{36}}\]
done
clear
D)
\[\frac{\text{8R}}{9}\]
done
clear
View Answer play_arrow
question_answer 41) In the nuclear reaction : \[^{\text{2}}\text{H}{{\text{e}}^{\text{4}}}{{\text{+}}_{\text{z}}}{{\text{X}}^{\text{A}}}{{\xrightarrow{{}}}_{\text{z+2}}}{{\text{Y}}^{\text{A+3}}}{{\text{+}}_{\text{z}}}{{\text{M}}^{\text{A }\!\!\!\!\text{ }}}\]where M denotes :
A)
electron
done
clear
B)
positron
done
clear
C)
proton
done
clear
D)
neutron
done
clear
View Answer play_arrow
question_answer 42) If the de-Broglie wavelength of a proton is\[{{10}^{-13}}m,\] the electric potential through which it must have been accelerated is :
A)
\[4.07\times {{10}^{4}}V\]
done
clear
B)
\[8.2\times {{10}^{4}}V\]
done
clear
C)
\[8.2\times {{10}^{3}}V\]
done
clear
D)
\[4.07\times {{10}^{5}}V\]
done
clear
View Answer play_arrow
question_answer 43) \[n\] alpha particles per second are emitted from N atoms of a radioactive element. The half-life of radioactive element is :
A)
\[\frac{n}{N}s\]
done
clear
B)
\[\frac{N}{n}s\]
done
clear
C)
\[\frac{0.693N}{n}s\]
done
clear
D)
\[\frac{0.693n}{N}s\]
done
clear
View Answer play_arrow
question_answer 44) Depletion layer consists of:
A)
electrons
done
clear
B)
protons
done
clear
C)
mobile ions
done
clear
D)
immobile ions
done
clear
View Answer play_arrow
question_answer 45) In a semiconductor diode the barrier potential offers opposition to only :
A)
majority carrier in both regions
done
clear
B)
minority carrier in both regions
done
clear
C)
free electrons in the \[n\]-region
done
clear
D)
holes in the \[p\]-region
done
clear
View Answer play_arrow
question_answer 46) Maximum entropy will be in which of the following?
A)
Ice
done
clear
B)
Liquid water
done
clear
C)
Snow
done
clear
D)
Water vapours
done
clear
View Answer play_arrow
question_answer 47) What is obtained when chlorine is passed in boiling toluene and product is hydrolysed?
A)
o-cresol
done
clear
B)
p-cresol
done
clear
C)
2, 4-dihydroxytoluene
done
clear
D)
Benzyl alcohol
done
clear
View Answer play_arrow
question_answer 48) Which of the following has covalent bond?
A)
\[N{{a}_{2}}S\]
done
clear
B)
\[AlC{{l}_{3}}\]
done
clear
C)
\[NaH\]
done
clear
D)
\[MgC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 49) Which of the following acts as an oxidising as well as reducing agent?
A)
\[N{{a}_{2}}O\]
done
clear
B)
\[N{{a}_{2}}{{O}_{2}}\]
done
clear
C)
\[NaN{{O}_{3}}\]
done
clear
D)
\[NaN{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 50) What is the oxidation state of P in \[Ba{{({{H}_{2}}P{{O}_{2}})}_{2}}\]?
A)
\[+1\]
done
clear
B)
\[+2\]
done
clear
C)
\[+3\]
done
clear
D)
\[-1\]
done
clear
View Answer play_arrow
question_answer 51) Which of the following molecules has pyramidal shape?
A)
\[PC{{l}_{3}}\]
done
clear
B)
\[S{{O}_{3}}\]
done
clear
C)
\[CO_{3}^{2-}\]
done
clear
D)
\[NO_{3}^{-}\]
done
clear
View Answer play_arrow
question_answer 52) Maximum number of -hydrogen bonding in is:
A)
\[1\]
done
clear
B)
\[2\]
done
clear
C)
\[3\]
done
clear
D)
\[4\]
done
clear
View Answer play_arrow
question_answer 53) Number of isomers possible for \[{{C}_{4}}{{H}_{8}}O\] is
A)
\[3\]
done
clear
B)
\[4\]
done
clear
C)
\[5\]
done
clear
D)
\[6\]
done
clear
View Answer play_arrow
question_answer 54) \[{{C}_{6}}{{H}_{5}}-CH=CHCHO\xrightarrow{X}\]\[{{C}_{6}}{{H}_{5}}CH=CHC{{H}_{2}}OH\] In the above sequence X can be:
A)
\[{{H}_{2}}/Ni\]
done
clear
B)
\[NaB{{H}_{4}}\]
done
clear
C)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}+{{H}^{+}}\]
done
clear
D)
both (a) and (b)
done
clear
View Answer play_arrow
question_answer 55) For a reaction \[{{H}_{2}}+{{I}_{2}}2HI\] at\[721K\], the value of equilibrium constant is 50. If \[~0.5\text{ }moles\] each of \[{{H}_{2}}\] and \[{{I}_{2}}\] is added to the system, the value of equilibrium constant will be:
A)
\[40\]
done
clear
B)
\[60\]
done
clear
C)
\[50\]
done
clear
D)
\[30\]
done
clear
View Answer play_arrow
question_answer 56) Schottky defect generally appears in:
A)
\[NaCl\]
done
clear
B)
\[KCl\]
done
clear
C)
\[CsCl\]
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 57) The ability of a given substance to assume two or more crystalline structure is called :
A)
amorphism
done
clear
B)
isomorphism
done
clear
C)
polymorphism
done
clear
D)
isomerism
done
clear
View Answer play_arrow
question_answer 58) A cricket ball of \[0.5kg\]is moving with a velocity of \[100m/sec\]. The wavelength associated with its motion is:
A)
\[1/100\text{ }cm\]
done
clear
B)
\[6.6\times {{10}^{-34}}m\]
done
clear
C)
\[1.32\times {{10}^{-35}}m\]
done
clear
D)
\[6.6\times {{10}^{-28}}m\]
done
clear
View Answer play_arrow
question_answer 59) Which among the following species have the same number of electrons in its outermost as well as penultimate shell?
A)
\[M{{g}^{2+}}\]
done
clear
B)
\[{{O}^{2-}}\]
done
clear
C)
\[{{F}^{-}}\]
done
clear
D)
\[C{{a}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 60) \[\Delta {{E}^{o}}\]of combustion of isobutylene is \[-X\text{ }kJ\text{ }mo{{l}^{-1}}\]. The value of \[\Delta {{H}^{o}}\] is:
A)
\[=\Delta {{E}^{o}}\]
done
clear
B)
\[>\Delta {{E}^{o}}\]
done
clear
C)
\[=0\]
done
clear
D)
\[<\Delta {{E}^{o}}\]
done
clear
View Answer play_arrow
question_answer 61) At \[{{90}^{o}}C\], pure water has \[{{H}_{3}}{{O}^{+}}\] ion concentration of \[{{10}^{-6}}mol/{{L}^{-1}}\]. The \[{{K}_{\omega }}\] at \[{{90}^{o}}C\] is:
A)
\[{{10}^{-6}}\]
done
clear
B)
\[{{10}^{-14}}\]
done
clear
C)
\[{{10}^{-12}}\]
done
clear
D)
\[{{10}^{-8}}\]
done
clear
View Answer play_arrow
question_answer 62) A gas is found to have a formula \[{{[CO]}_{x}}\]. If its vapour density is 70, the value of x is:
A)
\[2.5\]
done
clear
B)
\[3.0\]
done
clear
C)
\[5.0\]
done
clear
D)
\[6.0\]
done
clear
View Answer play_arrow
question_answer 63) Which of the following represents soap?
A)
\[{{C}_{17}}{{H}_{35}}COOK\]
done
clear
B)
\[{{C}_{17}}{{H}_{35}}COOH\]
done
clear
C)
\[{{C}_{15}}{{H}_{31}}COOH\]
done
clear
D)
\[{{({{C}_{17}}{{H}_{35}}COO)}_{2}}Ca\]
done
clear
View Answer play_arrow
question_answer 64) Aspirin in chemically:
A)
methyl benzoate
done
clear
B)
ethyl salicylate
done
clear
C)
acetyl salicylic acid
done
clear
D)
o-hydroxy benzoic acid
done
clear
View Answer play_arrow
question_answer 65) Vitamin \[{{B}_{6}}\] is known a:
A)
pyridoxin
done
clear
B)
thiamine
done
clear
C)
tocopherol
done
clear
D)
riboflavin
done
clear
View Answer play_arrow
question_answer 66) Which of the following compounds is found abundantly in nature?
A)
Fructose
done
clear
B)
Starch
done
clear
C)
Glucose
done
clear
D)
Cellulose
done
clear
View Answer play_arrow
question_answer 67) Synthetic polymer which resembles natural rubber is:
A)
neoprene
done
clear
B)
chloroprene
done
clear
C)
glyptal
done
clear
D)
nylon
done
clear
View Answer play_arrow
question_answer 68) Which of the. following is not a nitre-derivative
A)
\[{{C}_{6}}{{H}_{5}}N{{O}_{2}}\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}ONO\]
done
clear
C)
done
clear
D)
\[{{C}_{6}}{{H}_{4}}(OH)N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 69) The reduction of which of the following compound would yield secondary amine?
A)
Alkyl nitrile
done
clear
B)
Carbylamine
done
clear
C)
Primary amine
done
clear
D)
Secondary nitro compound
done
clear
View Answer play_arrow
question_answer 70) Which of the aldehyde is most reactive?
A)
\[{{C}_{6}}{{H}_{5}}-CHO\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
\[HCHO\]
done
clear
D)
All are equally reactive
done
clear
View Answer play_arrow
question_answer 71) Which of the following does not contain \[-COOH\] group?
A)
Aspirin
done
clear
B)
Benzoic acid
done
clear
C)
Picric acid
done
clear
D)
All have \[-COOH\] group
done
clear
View Answer play_arrow
question_answer 72) Which of the following is dihydric alcohol?
A)
Glycerol
done
clear
B)
Ethylene glycol
done
clear
C)
Catechol
done
clear
D)
Resorcinol
done
clear
View Answer play_arrow
question_answer 73) Ethyl alcohol is heated with cone. \[{{H}_{2}}S{{O}_{4}}\]. The product formed is:
A)
\[C{{H}_{3}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-O{{C}_{2}}{{H}_{5}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 74) In the first order reaction, the concentration of the reactants is reduced to 25% in one hour. The half-life period of the reaction is
A)
\[2h\]
done
clear
B)
\[4h\]
done
clear
C)
\[1/2h\]
done
clear
D)
\[1/4h\]
done
clear
View Answer play_arrow
question_answer 75) For a reaction \[X(g)\xrightarrow[{}]{}Y(g)+Z(g)\]the half-life period is 10 min. In what period of time would the concentration of X be reduced to 10% of original concentration?
A)
\[20\,min\]
done
clear
B)
\[33\,min\]
done
clear
C)
\[15\,min\]
done
clear
D)
\[25\,min\]
done
clear
View Answer play_arrow
question_answer 76) The molar freezing point constant for water is\[{{1.86}^{o}}C/mol\]. If 342 g of cane sugar \[({{C}_{12}}{{H}_{22}}{{O}_{11}})\]is dissolved in 1000 g of water, the solution will freeze at:
A)
\[-{{1.86}^{o}}C\]
done
clear
B)
\[{{1.86}^{o}}C\]
done
clear
C)
\[-{{3.92}^{o}}C\]
done
clear
D)
\[{{2.42}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 77) The movement of solvent molecules through a semipermeable membrane is called:
A)
Electrolysis
done
clear
B)
Electrophoresis
done
clear
C)
Osmosis
done
clear
D)
Cataphoresis
done
clear
View Answer play_arrow
question_answer 78) Which of the following is a primary halide?
A)
Isopropyl iodide
done
clear
B)
Secondary butyl iodide
done
clear
C)
Tertiary butyl bromide
done
clear
D)
Neo hexyl chloride
done
clear
View Answer play_arrow
question_answer 79) A gas is found to have the formula \[{{(CO)}_{n}}\]. If its vapour density is 56, the value of n will be:
A)
\[7\]
done
clear
B)
\[5\]
done
clear
C)
\[4\]
done
clear
D)
\[3\]
done
clear
View Answer play_arrow
question_answer 80) Aromatisation of n-heptane by passing over\[(A{{l}_{2}}{{O}_{3}}+C{{r}_{2}}{{O}_{3}})\] catalyst at \[773K\]gives:
A)
benzene
done
clear
B)
toluene
done
clear
C)
Mixture of both
done
clear
D)
Heptylene
done
clear
View Answer play_arrow
question_answer 81) \[{{C}_{6}}{{H}_{5}}C{{H}_{3}}\xrightarrow{Cr{{O}_{2}}\,C{{l}_{2}}}Z\]In the given sequence Z is:
A)
benzaldehyde
done
clear
B)
toluic acid
done
clear
C)
phenyl acetic acid
done
clear
D)
benzoic acid
done
clear
View Answer play_arrow
question_answer 82) Nitro ethane can exhibit one of the following kind of isomerism:
A)
metamerism
done
clear
B)
optical activity
done
clear
C)
tautomerism
done
clear
D)
position isomerism
done
clear
View Answer play_arrow
question_answer 83) A compound has 3 chiral carbon atoms. The number of possible optical isomers it can have:
A)
\[3\]
done
clear
B)
\[2\]
done
clear
C)
\[8\]
done
clear
D)
\[4\]
done
clear
View Answer play_arrow
question_answer 84) \[4{{K}_{2}}C{{r}_{2}}{{O}_{7}}\xrightarrow{heat}4{{K}_{2}}Cr{{O}_{4}}+3{{O}_{2}}+X\] heat In the above reaction X is:
A)
\[Cr{{O}_{3}}\]
done
clear
B)
\[C{{r}_{2}}{{O}_{7}}\]
done
clear
C)
\[C{{r}_{2}}{{O}_{3}}\]
done
clear
D)
\[Cr{{O}_{5}}\]
done
clear
View Answer play_arrow
question_answer 85) The co-ordination number and oxidation number of X in the following compound\[[X\,(S{{O}_{4}})\,{{(N{{H}_{3}})}_{5}}]Cl\]will be:
A)
\[10\] and \[3\]
done
clear
B)
\[2\] and \[6\]
done
clear
C)
\[6\] and \[3\]
done
clear
D)
\[6\] and \[4\]
done
clear
View Answer play_arrow
question_answer 86) In the electrolysis of water one faraday of electrical energy would evolve:
A)
\[1mole\]of oxygen
done
clear
B)
1 gram-atom of oxygen
done
clear
C)
8 g of oxygen
done
clear
D)
\[22.4\text{ }L\]of oxygen
done
clear
View Answer play_arrow
question_answer 87) In which of these process platinum is used as a catalyst?
A)
Oxidation of ammonia to form \[HN{{O}_{3}}\]
done
clear
B)
Hardening of oils
done
clear
C)
Production of synthetic rubber
done
clear
D)
Synthesis of methane
done
clear
View Answer play_arrow
question_answer 88) If the half-life of an isotope X is 10 years, its decay constant is:
A)
\[6.932\text{ }y{{r}^{-1}}\]
done
clear
B)
\[0.6932y{{r}^{-1}}\]
done
clear
C)
\[0.06932\text{ }y{{r}^{-1}}\]
done
clear
D)
\[0.006932\text{ }y{{r}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 89) On strong heating sodium bicarbonate changes into:
A)
sodium monoxide
done
clear
B)
sodium hydroxide
done
clear
C)
sodium carbonate
done
clear
D)
sodium peroxide
done
clear
View Answer play_arrow
question_answer 90) Which of the following is not an ore of magnesium?
A)
Carnallite
done
clear
B)
Magnesite
done
clear
C)
Dolomite
done
clear
D)
Gypsum
done
clear
View Answer play_arrow
question_answer 91) Sunnhemp is obtained from :
A)
Crotolaria juncea
done
clear
B)
Linum usitatiseimum
done
clear
C)
Corchorus capsularis
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 92) The enzyme involved in fixation of \[C{{O}_{2}}\] in \[{{C}_{4}}\] plants is :
A)
PEP carboxylase
done
clear
B)
RuDP carboxylase
done
clear
C)
RuDP oxydase
done
clear
D)
both (a) and (b)
done
clear
View Answer play_arrow
question_answer 93) Vegetative reproduction in Cycas occur by :
A)
bulbils
done
clear
B)
sporophylls
done
clear
C)
fission
done
clear
D)
scale leaves
done
clear
View Answer play_arrow
question_answer 94) Symbiotic relationship between blue-green algae and fungus is called :
A)
mycorrhiza
done
clear
B)
lichen
done
clear
C)
bryophyta
done
clear
D)
gymnosperm
done
clear
View Answer play_arrow
question_answer 95) Asafoetida (heeng) obtains from :
A)
root secretion
done
clear
B)
leaf
done
clear
C)
seed
done
clear
D)
fruit
done
clear
View Answer play_arrow
question_answer 96) Which plant hormone promotes seed, bud dormancy and causes stomatal closure?
A)
IAA
done
clear
B)
Abscisic acid
done
clear
C)
GAI
done
clear
D)
Cytokinin
done
clear
View Answer play_arrow
question_answer 97) Viscum album grows on trees. This is an example of:
A)
symbiosis
done
clear
B)
parasitism
done
clear
C)
commensalism
done
clear
D)
predation
done
clear
View Answer play_arrow
question_answer 98) In addition to absence of chlorophyll what is the other difference between fungi and higher plants?
A)
Type of nutrition and composition of cell wall
done
clear
B)
Cell type
done
clear
C)
Nucleus
done
clear
D)
Reproduction
done
clear
View Answer play_arrow
question_answer 99) Ozone layer is depleted by :
A)
\[S{{O}_{2}},\text{ }N{{O}_{3}}\]
done
clear
B)
\[CFC,\text{ }C{{H}_{4}},\text{ }{{N}_{2}}O\]
done
clear
C)
\[CO,\text{ }C{{H}_{4}},\text{ }{{\text{O}}_{2}}\]
done
clear
D)
\[N{{O}_{2}},\text{ }C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 100) Which is an electron carrier common to both photosynthesis and respiration?
A)
Cytochrome
done
clear
B)
Phytochrome
done
clear
C)
Phaophytin
done
clear
D)
Plastocynin
done
clear
View Answer play_arrow
question_answer 101) Hesperidium of orange is a modification of:
A)
berry
done
clear
B)
drupe
done
clear
C)
pome
done
clear
D)
aggregate fruit
done
clear
View Answer play_arrow
question_answer 102) Quinine used for treatment of malarial fever extrected from :
A)
Atropa belladona
done
clear
B)
Cinchona officinalis
done
clear
C)
Aconitum napellus
done
clear
D)
Rauwolffia serpentine
done
clear
View Answer play_arrow
question_answer 103) Larger nucleus in a pollen grain is :
A)
tube nucleus
done
clear
B)
sperm nucleus
done
clear
C)
generative nucleus
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 104) A pollutant can be best defined as it:
A)
has natural geochemical cycles
done
clear
B)
changes homeostasis of environment
done
clear
C)
disturb natural flora of a place
done
clear
D)
become stabilized in ecosystem forever
done
clear
View Answer play_arrow
question_answer 105) Enveloped virus enters into host cell by:
A)
injection own nucleic acid inside host cell
done
clear
B)
by contact with cell receptor and endocytosis
done
clear
C)
by phagocytosis
done
clear
D)
fusion with the plasma membrane of host
done
clear
View Answer play_arrow
question_answer 106) Most diverse organism of an ecosystem is :
A)
producer
done
clear
B)
consumer
done
clear
C)
decomposer
done
clear
D)
carnivores
done
clear
View Answer play_arrow
question_answer 107) Large size of pinae in animals of warm region in comparision to animal of cold region is due to law of:
A)
Dollos law
done
clear
B)
Glogers law
done
clear
C)
Copes law
done
clear
D)
Allens rule
done
clear
View Answer play_arrow
question_answer 108) Carthamus belong to family :
A)
Compositae
done
clear
B)
Gramineae
done
clear
C)
Liliaceae
done
clear
D)
Solanaceae
done
clear
View Answer play_arrow
question_answer 109) Restriction endonuclease cuts :
A)
one strand of DNA at specific site
done
clear
B)
both strand of DNA
done
clear
C)
both stands of DNA at any site
done
clear
D)
single strand of RNA
done
clear
View Answer play_arrow
question_answer 110) Photosynthetic products translocated in the form of:
A)
glucose
done
clear
B)
sucrose
done
clear
C)
maltose
done
clear
D)
starch
done
clear
View Answer play_arrow
question_answer 111) Raphides are crystals of:
A)
calcium carbonate
done
clear
B)
calcium oxalate
done
clear
C)
magnecium carbonate
done
clear
D)
magnecium oxalate
done
clear
View Answer play_arrow
question_answer 112) Number of meiosis for formation of 64 zygote in angiosperm is 80 but in gymnosperms number of meiosis for formation of 64 zygotes will:
A)
40
done
clear
B)
80
done
clear
C)
160
done
clear
D)
20
done
clear
View Answer play_arrow
question_answer 113) Catalytic converter in vehicle is used for controling :
A)
air pollution
done
clear
B)
water pollution
done
clear
C)
radioactive pollution
done
clear
D)
soil pollution
done
clear
View Answer play_arrow
question_answer 114) Monothecous anther is the characteristic of :
A)
Malvaceae
done
clear
B)
Liliaceae
done
clear
C)
Brassicaceae
done
clear
D)
Solanaceae
done
clear
View Answer play_arrow
question_answer 115) The onagrad type embryo development is found in:
A)
Solanum
done
clear
B)
Capsella
done
clear
C)
Lilium
done
clear
D)
Hibiscus
done
clear
View Answer play_arrow
question_answer 116) Rauwolfin obtains from which part of the plant?
A)
Stem
done
clear
B)
Root
done
clear
C)
Fruit
done
clear
D)
Leaf
done
clear
View Answer play_arrow
question_answer 117) Which type of placentation is -found in family Fabaceae?
A)
Axile
done
clear
B)
Marginal
done
clear
C)
Perietal
done
clear
D)
Basal
done
clear
View Answer play_arrow
question_answer 118) In meiotic division the DNA synthesis takes place in :
A)
S-phase
done
clear
B)
S-phase or leptotene phase
done
clear
C)
M-phase and zygotene
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 119) Thigmotropism occur due to the :
A)
light
done
clear
B)
contact
done
clear
C)
temperature
done
clear
D)
pressure
done
clear
View Answer play_arrow
question_answer 120) The maximum amount of calcium pectate is present in :
A)
primary cell wall
done
clear
B)
secondary cell wall
done
clear
C)
middle lamella
done
clear
D)
cell membrane
done
clear
View Answer play_arrow
question_answer 121) The special structure present in \[{{C}_{4}}\] plants is:
A)
thin cuticle
done
clear
B)
multi layered epidermis
done
clear
C)
kranz type body
done
clear
D)
one layered epidermis
done
clear
View Answer play_arrow
question_answer 122) White rust of Brassica is caused by :
A)
Albugo Candida
done
clear
B)
Ustilago
done
clear
C)
Phytophthora
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 123) Function of rough endoplasmic reticulum is :
A)
fat synthesis
done
clear
B)
protein synthesis
done
clear
C)
starch synthesis
done
clear
D)
autolysis
done
clear
View Answer play_arrow
question_answer 124) Which one of the following is a viral causing disease?
A)
Leaf curl of papaya
done
clear
B)
Red rot of sugarcane
done
clear
C)
Black rust of wheat
done
clear
D)
Late blight of potato
done
clear
View Answer play_arrow
question_answer 125) Tetradynamous androecium is found in :
A)
mustard
done
clear
B)
onion
done
clear
C)
tomato
done
clear
D)
sunflower
done
clear
View Answer play_arrow
question_answer 126) In mitochondria, enzymes responsible for Krebs cycle are present in:
A)
matrix
done
clear
B)
inner membrane
done
clear
C)
outer membrane
done
clear
D)
both (b) and (c)
done
clear
View Answer play_arrow
question_answer 127) In ferns and mosses, movement of antherozoids towards female component is called :
A)
phototaxis
done
clear
B)
chemotaxis
done
clear
C)
hydrotropism
done
clear
D)
thigmotropism
done
clear
View Answer play_arrow
question_answer 128) Plasmid is a :
A)
fungus
done
clear
B)
Plasrid
done
clear
C)
pan of plasma membrane
done
clear
D)
extra chromosomal DNA in bacterial cell
done
clear
View Answer play_arrow
question_answer 129) The reason for apical dominance is:
A)
ethylene
done
clear
B)
auxin
done
clear
C)
cytokinin
done
clear
D)
gibberellins
done
clear
View Answer play_arrow
question_answer 130) A plant bearing different type of leaves is called:
A)
heierophylly
done
clear
B)
heterotrophy
done
clear
C)
heterozygosity
done
clear
D)
heteronasty
done
clear
View Answer play_arrow
question_answer 131) Avena curvature test is a bioassay for examining the activity of:
A)
auxins
done
clear
B)
gibberellins
done
clear
C)
cytokinins
done
clear
D)
ethylene
done
clear
View Answer play_arrow
question_answer 132) What is common between chloroplast chromoplasts and leucoplasts?
A)
Presenece of pigments
done
clear
B)
Possession of thylakoids and grana
done
clear
C)
Storage of starch, proteins and lipids
done
clear
D)
Ability to multiply by a fission-like process
done
clear
View Answer play_arrow
question_answer 133) Plants of which one of the following groups of genera are pollinated by the same agency ?
A)
Triticum, mussanda, Zea mays
done
clear
B)
Kadam Cannabis
done
clear
C)
Salvia, Calotropis. Rafflesia
done
clear
D)
Salvia., Pinus, Ophrys
done
clear
View Answer play_arrow
question_answer 134) The montreal protocol refers to :
A)
persistent organic pollutants
done
clear
B)
global warming and climate change
done
clear
C)
substances that deplete the ozone layer
done
clear
D)
biosafety genetically modified organisms
done
clear
View Answer play_arrow
question_answer 135) Keystone species deserve protection because these :
A)
are capable of surviving in harsh environmental condition
done
clear
B)
indicate presence of certain minerals in the soil
done
clear
C)
have become rare due to over exploitation
done
clear
D)
play in important role in supporting other species
done
clear
View Answer play_arrow
question_answer 136) Chief cell of gastric gland are :
A)
simple tubular
done
clear
B)
coiled tubular
done
clear
C)
branched tubular
done
clear
D)
compound tubular
done
clear
View Answer play_arrow
question_answer 137) Antennae of cockroach function as :
A)
tactile and sensory receptor
done
clear
B)
gustatory receptor
done
clear
C)
auditor) recepror
done
clear
D)
olfactory receptor
done
clear
View Answer play_arrow
question_answer 138) Diabetes insipedus is caused by the deficiency of hormone:
A)
insulin
done
clear
B)
vasopressin
done
clear
C)
glucagon
done
clear
D)
oxytocin
done
clear
View Answer play_arrow
question_answer 139) Type of sexual reproduction in protists, bearing diploid chromosome is :
A)
zygotic meiosis
done
clear
B)
binary fission
done
clear
C)
cyst formation
done
clear
D)
gametangial meiosis
done
clear
View Answer play_arrow
question_answer 140) Glomerular area of adrenal cortex is responsible for:
A)
water and electrolyte balance
done
clear
B)
carbohydrate rpccabolism
done
clear
C)
steroid and hormone secretion
done
clear
D)
blood pressure
done
clear
View Answer play_arrow
question_answer 141) Down syndrome is due to:
A)
trisomy of 13th chromosome
done
clear
B)
non- disjunction of 21st chromosome
done
clear
C)
trisomy of 18th chromosome
done
clear
D)
fusion of normal sperm with egg having 2X
done
clear
View Answer play_arrow
question_answer 142) Marine teleost fishes excrete :
A)
uric acid
done
clear
B)
ammonia
done
clear
C)
urea
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 143) Part of nephron impermeable to salt:
A)
distal convoluted tubules
done
clear
B)
descending limb of loop of Henle
done
clear
C)
assending limb of loop of Henle
done
clear
D)
collecting duct
done
clear
View Answer play_arrow
question_answer 144) Nodes of Ranvier are found in :
A)
non myelinated nerve fibres
done
clear
B)
myelinated nerve fibres
done
clear
C)
both (a) and (b)
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 145) Myxoedema is a disorsder of:
A)
parathyroid gland
done
clear
B)
adrenal gland
done
clear
C)
pituitary gland
done
clear
D)
thyroid gland
done
clear
View Answer play_arrow
question_answer 146) SA node is located in :
A)
lower lateral wall of right atrium
done
clear
B)
upper lateral wall of right atrium
done
clear
C)
upper lateral wall of left atrium
done
clear
D)
lower kitcral wall of left atrium
done
clear
View Answer play_arrow
question_answer 147) Allergencs are:
A)
infectious and increases secretion of IgE
done
clear
B)
non infectious and increases secretions of IgE
done
clear
C)
infectious and increases secretion of IgG
done
clear
D)
noninfectious and increases secretion IgM
done
clear
View Answer play_arrow
question_answer 148) Mouth part of housefly are :
A)
siphoning type
done
clear
B)
sponging type
done
clear
C)
biting and chewing
done
clear
D)
piercing and sucking
done
clear
View Answer play_arrow
question_answer 149) Nissls granules are present in :
A)
RBC
done
clear
B)
WBC
done
clear
C)
Both (a) and (b)
done
clear
D)
nerve cells
done
clear
View Answer play_arrow
question_answer 150) Human dental formula is :
A)
\[\frac{3142}{3143}\]
done
clear
B)
\[\frac{1003}{0003}\]
done
clear
C)
\[\frac{2123}{2123}\]
done
clear
D)
\[\frac{2143}{3143}\]
done
clear
View Answer play_arrow
question_answer 151) Vitamin\[-{{B}_{12}}\] is available to ruminants by :
A)
plants
done
clear
B)
micro-organisms in caecum
done
clear
C)
animals
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 152) Which is symbiont inside human intestine?
A)
Streptococcus pneumonia
done
clear
B)
Neisseria meningitides
done
clear
C)
E. coli
done
clear
D)
Treponema pallidum
done
clear
View Answer play_arrow
question_answer 153) Human population growth is :
A)
lag
done
clear
B)
stationary
done
clear
C)
exponential
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 154) Maximum noise permissible during day time in residential areas :
A)
75 dB
done
clear
B)
55 dB
done
clear
C)
65 dB
done
clear
D)
45 dB
done
clear
View Answer play_arrow
question_answer 155) Trigeminal nerve m frog is of:
A)
4th
done
clear
B)
5th
done
clear
C)
8th
done
clear
D)
9th
done
clear
View Answer play_arrow
question_answer 156) Male and female cockroach is differs due to presence of absence of :
A)
anal cerci and anal style
done
clear
B)
anal cerci
done
clear
C)
anal style
done
clear
D)
anal sitae
done
clear
View Answer play_arrow
question_answer 157) Addison of which elements in water speed up the metamorphosis in frog tadpole larva?
A)
\[{{\text{I}}_{\text{2}}}\]
done
clear
B)
K
done
clear
C)
Na
done
clear
D)
Cl
done
clear
View Answer play_arrow
question_answer 158) Adrenaline and nor-adrenaline are hormones and acts as :
A)
energy producing agent
done
clear
B)
food storage material
done
clear
C)
neuro-transmitter
done
clear
D)
energy storing substances
done
clear
View Answer play_arrow
question_answer 159) Largest muscle in the human body is :
A)
sartorius
done
clear
B)
gluteus
done
clear
C)
stapedius
done
clear
D)
masseter
done
clear
View Answer play_arrow
question_answer 160) Blood vascular system of earthworm is :
A)
open type with Hb in RBC
done
clear
B)
open type with Hb in plasma
done
clear
C)
closed type with Hb in RBC
done
clear
D)
closed type with Hb in plasma
done
clear
View Answer play_arrow
question_answer 161) Which types of cells absent in sponges?
A)
Trophocytes
done
clear
B)
Myocytes
done
clear
C)
Archeocytes
done
clear
D)
Cnidocytes
done
clear
View Answer play_arrow
question_answer 162) Third moulting in Ascari.s larva takes place in :
A)
lung
done
clear
B)
liver
done
clear
C)
heart
done
clear
D)
intestine
done
clear
View Answer play_arrow
question_answer 163) Liver and pancreas is derivative of:
A)
ectoderm
done
clear
B)
endoderm
done
clear
C)
ectoderm and mesoderm
done
clear
D)
ectoderm and endoderm
done
clear
View Answer play_arrow
question_answer 164) An important evidence in favour of organic evolution is the occurrence of:
A)
homologous and vestigial organs
done
clear
B)
analogous and vestigial organs
done
clear
C)
homologous organs only
done
clear
D)
homologous and analogous organs
done
clear
View Answer play_arrow
question_answer 165) Which one of the following is not a living fossil?
A)
King crab
done
clear
B)
Sphenodon
done
clear
C)
Archaeopteryx
done
clear
D)
Peripatus
done
clear
View Answer play_arrow
question_answer 166) Which one of the following is a matching set of a phylum and its three examples?
A)
Cnidaria - Bonellia, Physalia, Aurelia
done
clear
B)
Platyhelminthes - Planaria, Schistosoma, Enterobius
done
clear
C)
Mollusca - Loligo, Teredo, Octopus
done
clear
D)
Porifera - Spongilla, Euplectella, Pennatula
done
clear
View Answer play_arrow
question_answer 167) In Amoeba, osmoregulation takes place by :
A)
contractile vacuole
done
clear
B)
ectoplasm
done
clear
C)
pseudopodia
done
clear
D)
hyaloplasm
done
clear
View Answer play_arrow
question_answer 168) The infective stage ofPlasmodium in human is:
A)
sporozoite
done
clear
B)
trophozoite
done
clear
C)
gametocyte
done
clear
D)
merozoite
done
clear
View Answer play_arrow
question_answer 169) Chitin as exoskeleton found in :
A)
Periplaneta
done
clear
B)
Ascaris
done
clear
C)
Pheretima
done
clear
D)
Hydra
done
clear
View Answer play_arrow
question_answer 170) Pseudocoel is present in :
A)
Periplaneta
done
clear
B)
Ascaris
done
clear
C)
Pheretima
done
clear
D)
Hydra
done
clear
View Answer play_arrow
question_answer 171) Metameric segmentation is the main feature of:
A)
Annelida
done
clear
B)
Echinodermata
done
clear
C)
Arthropoda
done
clear
D)
Coelenterata
done
clear
View Answer play_arrow
question_answer 172) Nemaroblasts are formed by ;
A)
interstitial cells
done
clear
B)
glands cells
done
clear
C)
mesoepithelial cells
done
clear
D)
nerve cells
done
clear
View Answer play_arrow
question_answer 173) Which is not correct for birds?
A)
Exothermic
done
clear
B)
Pneumatic bones
done
clear
C)
Lung with air sacs
done
clear
D)
Amniotes
done
clear
View Answer play_arrow
question_answer 174) Which character is not same in Aves and mammals?
A)
Single systemic arch
done
clear
B)
Metanephric kidney
done
clear
C)
Seven cervical vertebrae
done
clear
D)
Homeotherms
done
clear
View Answer play_arrow
question_answer 175) Solenocytes and nephridia are respectively found in :
A)
Platyhelminth and Annelids
done
clear
B)
Annelids and Nematoda
done
clear
C)
Cendaria and Mollusca
done
clear
D)
Mollusca and Echinodermata
done
clear
View Answer play_arrow
question_answer 176) Post ganglionic nerve fibres of sympathetic system are :
A)
adrenergic
done
clear
B)
cholinergic
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 177) Which is not used up in human body?
A)
Calcium
done
clear
B)
Phosphorus
done
clear
C)
Zinc
done
clear
D)
Barium
done
clear
View Answer play_arrow
question_answer 178) Ichthyophis belongs to:
A)
Mammalia
done
clear
B)
Reptilia
done
clear
C)
Amphibia
done
clear
D)
Aves
done
clear
View Answer play_arrow
question_answer 179) The supporting and nutritive cells found in the brain are :
A)
ependymal cells
done
clear
B)
microglia
done
clear
C)
astrocytes
done
clear
D)
oligodendrocytes
done
clear
View Answer play_arrow
question_answer 180) Prolonged deficiency of thiamine in human diet may lead to :
A)
pellegra
done
clear
B)
beri-beri
done
clear
C)
anaemia
done
clear
D)
Haemorrhage
done
clear
View Answer play_arrow