Answer:
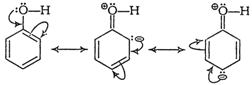
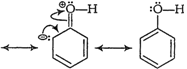
In the presence of attacking electrophile, ?OH group
exerts+R effect on the benzene ring. So, electron density in the ring
increasesparticularly at the ortho and para positions. When an
electrophileattacks, substitution takes place readily at these (oandp-)
positions.
You need to login to perform this action.
You will be redirected in
3 sec
