Answer:
Ans. (i) [Cr(C2O4)3]3-.
No geometrical isomerism is shown.
(ii) Two isomers
are possible for [Co(NH3)3Cl3]
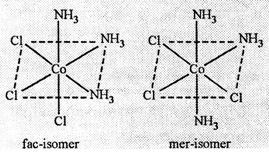 Note. 1, 2, 3,-position
is called cis isomer while position 1, 2, 6 is called as trans isomer.
In cis-form,
similar groups occupy the corners of one of the octahedral faces and hence is
called facial or face-isomer. On the other hand, in trans-form the positions
are around the meridian of the octahedron, hence is called meridional (mer)
isomer.
Note. 1, 2, 3,-position
is called cis isomer while position 1, 2, 6 is called as trans isomer.
In cis-form,
similar groups occupy the corners of one of the octahedral faces and hence is
called facial or face-isomer. On the other hand, in trans-form the positions
are around the meridian of the octahedron, hence is called meridional (mer)
isomer.
You need to login to perform this action.
You will be redirected in
3 sec
