Answer:
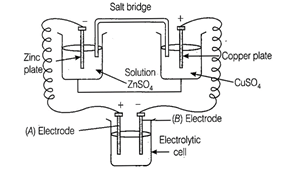
The cell drawn above represents
electrochemical cell in which two different electrodes are fitted in their
respective electrolytic solution and cell drawn at bottom represents electrolytic
cell.
Cell representation can be represented
as ![]() Zn is loosing electrons which
are going towards electrode (A) and copper is accepting electron from electrode
B. Hence,
Charge on electrode A = +
Charge on electrode B = -
Zn is loosing electrons which
are going towards electrode (A) and copper is accepting electron from electrode
B. Hence,
Charge on electrode A = +
Charge on electrode B = -
You need to login to perform this action.
You will be redirected in
3 sec
