Answer:
Due
to resonance, C?X bond in haloarenes and haloalkenes have some double bond
character but in
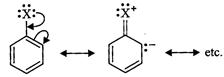
![]() haloalkanes, it is a pure single bond.
In other words, the C?X bond in haloarenes and haloalkenes is little
stronger than in haloalkanes and hence little more difficult to break. Thus, haloarenes
and haloalkenes are less reactive than haloalkanes.
haloalkanes, it is a pure single bond.
In other words, the C?X bond in haloarenes and haloalkenes is little
stronger than in haloalkanes and hence little more difficult to break. Thus, haloarenes
and haloalkenes are less reactive than haloalkanes.
You need to login to perform this action.
You will be redirected in
3 sec
