Answer:
The
given reaction is an example of electrophilic addition reaction in which a
proton adds first to form a carbocation which is then attacked by ![]()
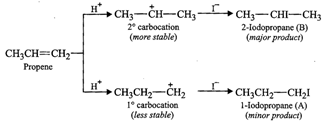 Since 2° carbocation is more stable than 1° carbocation, therefore, 2-iodopropane
(B) is the major product and 1-iodopropane (A) is the minor product.
The same conclusion can be drawn from Markovnikov 's rule which states
that the major product is obtain by addition of the negative part of the adding
molecule to that carbon atom of the double bond which carries lesser number of
hydrogen atom. Thus, 2-iodopropane (B) is the major product while l-iodoproane is
the minor product.
Since 2° carbocation is more stable than 1° carbocation, therefore, 2-iodopropane
(B) is the major product and 1-iodopropane (A) is the minor product.
The same conclusion can be drawn from Markovnikov 's rule which states
that the major product is obtain by addition of the negative part of the adding
molecule to that carbon atom of the double bond which carries lesser number of
hydrogen atom. Thus, 2-iodopropane (B) is the major product while l-iodoproane is
the minor product.
You need to login to perform this action.
You will be redirected in
3 sec
