Answer:
(i)
Since the rate of reaction of compound 'A' with aq. KOH depends upon the
concentration
of compound 'A' only, therefore, the reaction occurs by ![]() mechanism and the compound 'A'
2-bromo-2- methylpropane or tert-butyl bromide.
mechanism and the compound 'A'
2-bromo-2- methylpropane or tert-butyl bromide.
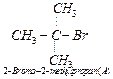
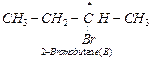 (ii) Since compound 'B' is optically active and is an isomer of compound
'A' with MF C4HgBr, therefore, compound 'B' must be 2-bromobutane. Further,
since the rate of reaction of compound 'B' with aq. KOH depends both' upon the
concentration of compound 'B' and KOH, therefore, the reaction occurs by
(ii) Since compound 'B' is optically active and is an isomer of compound
'A' with MF C4HgBr, therefore, compound 'B' must be 2-bromobutane. Further,
since the rate of reaction of compound 'B' with aq. KOH depends both' upon the
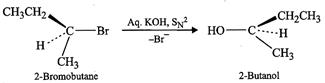
concentration of compound 'B' and KOH, therefore, the reaction occurs by ![]() mechanism. Since in
mechanism. Since in ![]() reactions, nucleophile attacks
from the back side, therefore, the product of hydrolysis
(i.e. 2-butanol) will have inverted configuration w.r.t. to
2-bromobutane.
reactions, nucleophile attacks
from the back side, therefore, the product of hydrolysis
(i.e. 2-butanol) will have inverted configuration w.r.t. to
2-bromobutane.
You need to login to perform this action.
You will be redirected in
3 sec
