Answer:
The
hydrocarbon having molecular mass of 72 g ![]() must
be
must
be ![]() Since on photochlorination, it
gives a single monochloro derivative, therefore, all the twelve hydrogens in
this hydrocarbon are equivalent.
Thus, the hydrocarbon must be neopentane or 2,2-dimethylpropane. It forms
only one monochloro derivative and two dichloro derivatives as shown below :
Since on photochlorination, it
gives a single monochloro derivative, therefore, all the twelve hydrogens in
this hydrocarbon are equivalent.
Thus, the hydrocarbon must be neopentane or 2,2-dimethylpropane. It forms
only one monochloro derivative and two dichloro derivatives as shown below :

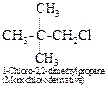
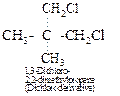
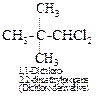
You need to login to perform this action.
You will be redirected in
3 sec
