Answer:
Catalyst is used to increase rate of
reaction. Heterogeneous catalyst is used to increase rate of reaction in which
catalyst is not in phase with reactants and products.
Role of adsorption in heterogeneous
catalysis are
(i) Diffusion of reactants to
the surface of the catalyst.
(ii) Reactants are adsorbed on
the catalyst surface.
(iii) Occurrence of chemical
reaction at catalyst surface.
(iv) Desorption.
(v) Diffusion of reaction
products away from the cataiysts surface.
e.g., 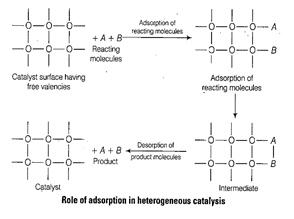
You need to login to perform this action.
You will be redirected in
3 sec
