Answer:
The graph between amount
of gas adsorbed per unit mass of the adsorbent x /m and pressure of the gas at
a constant temperature is known as Adsorption Isotherm. After saturation
pressure there is no more adsorption on the surface.
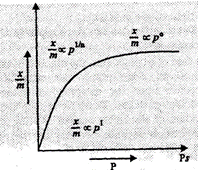 At low pressure:
At low pressure:
![]() is directly
proportional to pressure
is directly
proportional to pressure
![]()
![]() ?
(i)
where k is a
constant of proportionality.
At high pressure:
The value of
?
(i)
where k is a
constant of proportionality.
At high pressure:
The value of ![]() remains
same indicating that
remains
same indicating that ![]() is independent of
pressure.
is independent of
pressure.
![]() ?. (ii)
At intermediate
pressure:
The value of
?. (ii)
At intermediate
pressure:
The value of ![]() is proportional to
some fractional power of pressure
is proportional to
some fractional power of pressure
![]() ??. (iii)
where n = 1, 2, 3,
4. ..
The equation (iii)
is called Freundlich adsorption isotherm.
??. (iii)
where n = 1, 2, 3,
4. ..
The equation (iii)
is called Freundlich adsorption isotherm.
You need to login to perform this action.
You will be redirected in
3 sec
