Answer:
Conductivity of a semiconductor is too low
for practical use. The conductivity of a semiconductor can be increased by
adding a suitable amount of impurity to perfect crystal. This process is known
as doping. It can be done by adding either of two types of impurity to the
crystal.
(A) By adding electron rich impurities
i.e., group 15 elements to the silicon and germanium of group 14 elements. Out
of 4 valence electrons of group 14 elements and 5 valence electrons of group 15
elements, four electrons of each element led to formation of four covalent
bonds while the one extra electron of group 15?? elements become delocalised.
Thus, increases conductivity of
semiconductor. This type of semiconductor is known as n-type semiconductor.
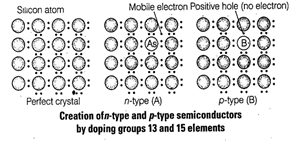 (B) By adding electron deficient
impurity i.e., group 14 to the perfect crystal of group 14 elements when group
13 element is doped to group 14 element it lead to create .a hole in the ideal
crystal which is known as electron hole or electron vacancy.
An electron from the
neighbouring atom come and fill the electron hole in doing so an electron from
the neighbour leaves an electron hole to its original position. Thus, it increases
conductivity of semiconductor. This type of semiconductor is known as p-type semiconductor.
(B) By adding electron deficient
impurity i.e., group 14 to the perfect crystal of group 14 elements when group
13 element is doped to group 14 element it lead to create .a hole in the ideal
crystal which is known as electron hole or electron vacancy.
An electron from the
neighbouring atom come and fill the electron hole in doing so an electron from
the neighbour leaves an electron hole to its original position. Thus, it increases
conductivity of semiconductor. This type of semiconductor is known as p-type semiconductor.
You need to login to perform this action.
You will be redirected in
3 sec
