Mass, Weight and Density
Category : Railways
Mass, Weight and Density
Mass
The mass (m) of a body of matter is quantitative measure of its inertia i.e., its resistance to a change in the state of rest or motion of the body, when a force is applied.
Inertia is the property of a mass which resists change from its states of rest or motion.
Volume (V) is defined as the amount of space occupied by a three-dimensional object as measured in cubic units.
Weight
The weight of an object is defined as the force of gravity on the object and may be calculated as the mass times the acceleration of gravity, w = mg. Since the weight is a force, its SI unit is the
Newton.
For an object in free fall, so that gravity is the only force acting on it,
Then the expression for weight follows from Newton's second law.
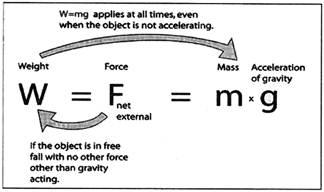
The value of g allows us to determine the net gravity force if it were in freefall and that net gravity force is the weight. Another approach is to consider "g" to be the measure of the intensity of
Ac gravity field in Newtons/kg at our location. We can view the weight as a measure of the mass in kg times the intensity of the gravity field, 9.8 Newton's/kg under standard conditions.
Density
Density (p) is defined as the mass of a substance per unit volume.
There are two kinds of density, "weight density" and "mass density". We will only use mass density and when we say: "density", its means "mass density".
The metric system was designed so that water will have a density of one gram per cubic centimeter or 1000 kilograms per cubic meter. Lead is about 10 times as dense as water and Styrofoam is about one tenth as dense as water.
Fluid Density
Mass per unit volume is defined as density. So density at a point of a fluid is represented as
\[\rho =\underset{\Delta V\to 0}{\mathop{\lim }}\,\frac{\Delta m}{\Delta V}=\frac{dm}{dV}\]
where m is the mass and v is the volume of the fluid. Density is a positive scalar quantity.
SI unit: \[kg/{{m}^{3}}\]
Dimensions: \[[M{{L}^{-3}}{{T}^{0}}]\]
i.e.\[{{M}_{body}}={{M}_{sub}}\]then\[{{V}_{body}}={{V}_{sub}}\]and\[{{\rho }_{body}}={{\rho }_{sub}}.\]
But for a hollow body or body with air gaps
\[{{M}_{body}}={{M}_{sub}}\]and\[{{V}_{body}}>{{V}_{sub}}\]then\[{{\rho }_{body}}<{{\rho }_{sub}}\]
\[{{M}_{MIX}}={{m}_{1}}+{{m}_{2}}\]and\[{{V}_{MIX}}={{V}_{1}}+{{V}_{2}}=\]\[\frac{{{m}_{1}}}{{{\rho }_{1}}}+\frac{{{m}_{2}}}{{{\rho }_{2}}}\]
\[\therefore \] \[\rho \,\,mix=\frac{M\,\,mix}{V\,\,mix}=\frac{{{m}_{1}}+{{m}_{2}}}{\underset{\rho 1}{\mathop{\underline{{{m}_{1}}}}}\,+\underset{\rho 2}{\mathop{\underline{{{m}_{2}}}}}\,}\]
\[\rho \,\,mix=\frac{2{{\rho }_{1}}{{\rho }_{2}}}{{{\rho }_{1}}+{{\rho }_{2}}}\](harmonic mean of individual densities)
\[{{V}_{MIX}}={{V}_{1}}+{{V}_{2}}\] and,
\[{{M}_{MIX}}={{m}_{1}}+{{m}_{2}}={{\rho }_{1}}{{V}_{1}}+{{\rho }_{2}}{{V}_{2}}\]
\[\therefore \] \[\rho \,\,mix=\frac{M\,\,mix}{V\,\,mix}=\frac{{{\rho }_{1}}{{V}_{1}}+{{\rho }_{2}}{{V}_{2}}}{{{V}_{1}}+{{V}_{2}}}\]
\[\rho \,\,mix=\frac{{{\rho }_{1}}+{{\rho }_{2}}}{2}\](arithmetic mean of individual densities)
Specific Weight or Weight Density (W)
It is defined as the ratio of the weight of the fluid to its volume or the weight acting per unit volume of the fluid.
Specific weight, \[W=\frac{Weight}{Volume}\]
\[W=\frac{mg}{V}=\left[ \frac{m}{V} \right]g=\rho g\]
S I Unit:\[N/{{m}^{3}}\] Dimensions:\[[M{{L}^{-2}}{{T}^{-2}}]\]
Specific weight of pure water at \[4{}^\circ C\] is \[9.81\,\,kN/{{m}^{3}}\]
Relative Density
It is defined as the ratio of the density of the given fluid to the density of pure water at \[4{}^\circ C\]
Density of given liquid
Relative density (R.D).\[=\frac{Density\,\,of\,\,given\,\,liquid}{Density\,\,of\,\,pure\,\,water\,\,at\,\,4{}^\circ C}\]
The density of water is maximum at \[4{}^\circ C\] and is equal to \[1.0\times {{10}^{3}}kg{{m}^{-3}}\]
Relative density or specific gravity is a unitless and dimensionless positive scalar physical quantity.
Being a dimensionless/unitless quantity R.D. of a substance is same in SI and CGS system.
Specific Gravity
It is defined as the ratio of the specific weight of the given fluid to the specific weight of pure water at\[4{}^\circ C\],
Specific gravity
\[=\frac{Specific\,\,weight\,\,of\,\,given\,liquid}{Specific\,\,weight\,\,of\,\,pure\,\,water\,\,at\,\,4{}^\circ C(9.81kN/{{m}^{3}})}\]
\[=\frac{{{\rho }_{\gamma }}\times g}{{{\rho }_{w}}\times g}=\frac{{{\rho }_{\gamma }}}{{{\rho }_{w}}}\]=R.D. of the liquid
Thus specific gravity of a liquid is numerically equal to the relative density of that liquid and for calculation purposes they are used interchangeably.
You need to login to perform this action.
You will be redirected in
3 sec
