Answer:
(i) Roasting: It is the process in
which sulphide ores of the metals are converted into oxides by heating them in
the presence of excess air. For example, zinc sulphide is converted into zinc
oxide by roasting.
2ZnS(s) +3O2(g) ![]() 2ZnO(s) +
2SO2(g)
Calcination:
It is the process in which carbonate ores of the metals are decomposed into
oxides by heating them in the absence or limited air. For example, zinc
carbonate is decomposed into zinc oxide and carbon dioxide by calcination.
ZnCO3O(s)
2ZnO(s) +
2SO2(g)
Calcination:
It is the process in which carbonate ores of the metals are decomposed into
oxides by heating them in the absence or limited air. For example, zinc
carbonate is decomposed into zinc oxide and carbon dioxide by calcination.
ZnCO3O(s) ![]() ZnO(s) +
CO2(g)
Out
of roasting and calcination, only roasting is used for sulphide ores. This is
because it is easier to obtain metal from its oxide as compared to its sulphide.
(ii)
ZnO(s) +
CO2(g)
Out
of roasting and calcination, only roasting is used for sulphide ores. This is
because it is easier to obtain metal from its oxide as compared to its sulphide.
(ii)
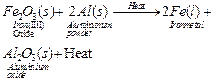 (iii) Anode ? Impure copper
Cathode ? Strip of pure copper
Electrolyte
? Acidified copper sulphate solution.
(iii) Anode ? Impure copper
Cathode ? Strip of pure copper
Electrolyte
? Acidified copper sulphate solution.
You need to login to perform this action.
You will be redirected in
3 sec
