Answer:
Cresol is methyl phenol. It has
three positional isomers : o-cresol, m-cresol and p-cresol.
Due to \[+I\]-effect of \[C{{H}_{3}}\] group,
electron-density in the \[O-H\] bond increases. Therefore, it becomes difficult
to break the \[O-H\] bond as compared to that in phenol and hence all cresols
are less acidic than phenol or phenol is a stronger acid than cresols.
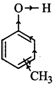 (Due to \[+I\]-effect of \[C{{H}_{3}}\]
group, electron density in the \[O-H\] bond increases thereby making the
cleavage of \[O-H\] bond difficult)
(Due to \[+I\]-effect of \[C{{H}_{3}}\]
group, electron density in the \[O-H\] bond increases thereby making the
cleavage of \[O-H\] bond difficult)
You need to login to perform this action.
You will be redirected in
3 sec
