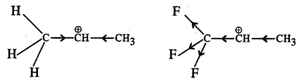
Answer:
\[C{{F}_{3}}\overset{\oplus
}{\mathop{C}}\,HC{{H}_{3}}\]more reactive because F atom being highly
electronegative has a strong-\[I\] effect. It destabilises the carbocation
towards nucleophilic attack as compared to \[C{{H}_{3}}\overset{\oplus
}{\mathop{C}}\,HC{{H}_{3}}\] in which \[C{{H}_{3}}\] group is an electron
releasing group and it stabilises the carbocation.
You need to login to perform this action.
You will be redirected in
3 sec
