Answer:
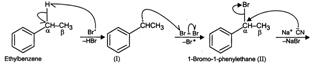
When ethyl benzene is treated with in
presence of light, the bromine free radical formed during the reaction,
abstracts the \[\alpha \] (but not \[\beta \]-)hydrogen from the ethyl group to
form a resonance stabilized benzylic radical \[(I)\] which then reacts with \[B{{r}_{2}}\]
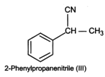
to form 1-bromo-1-phenylethane\[(II)\]. This on treatment with \[NaCN\]undergoes
nucleophilic substitution to form 2-phenylpropanenitrile \[(III)\].
You need to login to perform this action.
You will be redirected in
3 sec
