Answer:
The structures of all the possible alkyl halides that can be obtained by monohalogenation of 2-methylbutane are :
\[\underset{\text{1-Bromo-3-methylbutane}}{\mathop{C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{\overset{3}{\mathop{C}}\,}}\,H-C{{H}_{2}}\overset{1}{\mathop{C}}\,{{H}_{2}}Br}}\,\] \[\underset{\text{2-Bromo-3-methylbutane}}{\mathop{C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{\overset{3}{\mathop{\,\,CH}}\,}}\,-\underset{\begin{smallmatrix} | \\ Br \end{smallmatrix}}{\mathop{\overset{\,\,\,\,\,\,\,\,\,2}{\mathop{\,\,\,C}}\,H}}\,-C{{H}_{3}}}}\,\] \[\underset{\text{1-Bromo-2methylbutane}}{\mathop{Br-\overset{1}{\mathop{C}}\,{{H}_{2}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{\overset{2}{\mathop{C}}\,}}\,H-C{{H}_{2}}C{{H}_{3}}}}\,\] 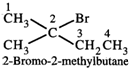 The Grignard reagents of all these four alkyl halides will give 2-methylbutane on treatment with ethanol.
The Grignard reagents of all these four alkyl halides will give 2-methylbutane on treatment with ethanol.
You need to login to perform this action.
You will be redirected in
3 sec
