Answer:
(i) \[3{}^\circ \]Alkyl halides on treatment with a strong base undergo dehydrohalogenation to form alkenes, i.e.,
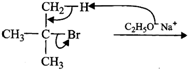
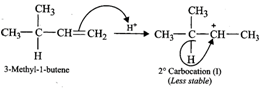
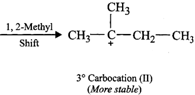
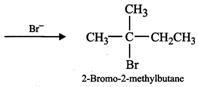
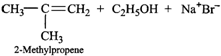 (ii) Addition of \[{{H}^{+}}\] to 3-methyl-1-butene first gives \[2{}^\circ \] carbocation \[(I)\] which being less stable rearranges to the more stable \[3{}^\circ \] carbocation \[(II)\]. Nucleophilic attack by \[B{{r}^{-}}\] ion on this carbocation gives 2-bromo-2-methylbutane as the major product,
(ii) Addition of \[{{H}^{+}}\] to 3-methyl-1-butene first gives \[2{}^\circ \] carbocation \[(I)\] which being less stable rearranges to the more stable \[3{}^\circ \] carbocation \[(II)\]. Nucleophilic attack by \[B{{r}^{-}}\] ion on this carbocation gives 2-bromo-2-methylbutane as the major product,
You need to login to perform this action.
You will be redirected in
3 sec
