-
question_answer1)
The end product in the following sequence of reaction is \[HC\equiv CH\underset{20%{{H}_{2}}S{{O}_{4}}}{\mathop{\xrightarrow{\text{1 }\!\!%\!\!\text{ HgS}{{\text{O}}_{\text{4}}}}}}\,A\xrightarrow{C{{H}_{3}}MgX}B\xrightarrow{[O]}\] [Bihar CEE 2002]
A)
Acetic acid done
clear
B)
Isopropyl alcohol done
clear
C)
Acetone done
clear
D)
Ethanol done
clear
View Solution play_arrow
-
question_answer2)
In the following reaction, product P is \[R-\overset{O\,\,\,}{\mathop{\overset{||\,\,\,\,}{\mathop{C-}}\,}}\,Cl\] \[\underset{Pd-BaS{{O}_{4}}}{\mathop{\xrightarrow{{{H}_{2}}}}}\,P\] [CBSE PMT 1991, 2000, 02; Kerala CET 2001; IIT 1992; AIIMS 1997; AFMC 1998]
A)
\[RC{{H}_{2}}OH\] done
clear
B)
\[R\,COOH\] done
clear
C)
\[RCHO\] done
clear
D)
\[RC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer3)
Acetophenone is prepared from [CPMT 2003]
A)
Rosenmund reaction done
clear
B)
Sandmayer reaction done
clear
C)
Wurtz reaction done
clear
D)
Friedel craft reaction done
clear
View Solution play_arrow
-
question_answer4)
Compound which gives acetone on ozonolysis [UPSEAT 2003]
A)
\[C{{H}_{3}}-CH=CH-C{{H}_{3}}\] done
clear
B)
\[{{(C{{H}_{3}})}_{2}}C=C{{(C{{H}_{3}})}_{2}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}CH=C{{H}_{2}}\] done
clear
D)
\[C{{H}_{3}}CH=C{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer5)
\[C{{H}_{3}}-\underset{\underset{O}{\mathop{||}}\,}{\mathop{C}}\,-C{{H}_{2}}-COO{{C}_{2}}{{H}_{5}}\underset{{{H}_{2}}O}{\mathop{\xrightarrow{NaOH}}}\,A,\] product ?A? in the reaction is [RPMT 2003]
A)
\[C{{H}_{3}}COOH\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
C)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}CHO\] done
clear
View Solution play_arrow
-
question_answer6)
Which one of the following compounds is prepared in the laboratory from benzene by a substitution reaction [EAMCET 2003]
A)
Glyoxal done
clear
B)
Cyclohexane done
clear
C)
Acetophenone done
clear
D)
Hexabromo cyclohexane done
clear
View Solution play_arrow
-
question_answer7)
Ketones \[(R-\underset{\underset{O}{\mathop{||}}\,}{\mathop{C}}\,-{{R}_{1}})\] where \[R={{R}_{1}}=\] alkyl group. It can be obtained in one step by [CBSE PMT 1997]
A)
Hydrolysis of esters done
clear
B)
Oxidation of primary alcohol done
clear
C)
Oxidation of secondary alcohol done
clear
D)
Reaction of acid halide with alcohols done
clear
View Solution play_arrow
-
question_answer8)
Predict the product ?B? in the sequence of reaction \[HC\equiv CH\underset{HgS{{O}_{4}}}{\mathop{\xrightarrow{30%{{H}_{2}}S{{O}_{4}}}}}\,A\xrightarrow{NaOH}B\] [CBSE PMT 2001]
A)
\[C{{H}_{3}}COONa\] done
clear
B)
\[C{{H}_{3}}COOH\] done
clear
C)
\[C{{H}_{3}}CHO\] done
clear
D)
\[C{{H}_{3}}-\underset{OH\,\,}{\mathop{\underset{|\,\,\,\,\,\,\,\,\,\,}{\mathop{CH-}}\,}}\,C{{H}_{2}}CHO\] done
clear
View Solution play_arrow
-
question_answer9)
\[C{{H}_{3}}COCl\underset{Pd/BaS{{O}_{4}}}{\mathop{\xrightarrow{2H}}}\,C{{H}_{3}}CHO+HCl;\] The above reaction is called [JIPMER 1997]
A)
Reimer-Tiemann reaction done
clear
B)
Cannizzaro reaction done
clear
C)
Rosenmund reaction done
clear
D)
Reformatsky reaction done
clear
View Solution play_arrow
-
question_answer10)
The oxidation of toluene to benzaldehyde by chromyl chloride is called [CBSE PMT 1996; AFMC 1998, 99; AIIMS 2000; JIPMER 2001; AFMC 2001; DCE 2004]
A)
Cannizzaro reaction done
clear
B)
Wurtz reaction done
clear
C)
Etard reaction done
clear
D)
Reimer-Tiemann reaction done
clear
View Solution play_arrow
-
question_answer11)
From which of the following tertiary butyl alcohol is obtained by the action of methyl magnesium iodide [MP CET 2000]
A)
\[HCHO\] done
clear
B)
\[C{{H}_{3}}CHO\] done
clear
C)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
D)
\[C{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer12)
Catalyst used in Rosenmund reduction is [Bihar MEE 1997]
A)
Pd / \[BaS{{O}_{4}}\] done
clear
B)
Zn-Hg couple done
clear
C)
\[LiAl{{H}_{4}}\] done
clear
D)
\[Ni/{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer13)
\[C{{H}_{3}}-C{{H}_{2}}-C\equiv CH\underset{{{H}_{2}}O}{\mathop{\xrightarrow{R}}}\,\]Butanone, R is [BHU 2003]
A)
\[H{{g}^{++}}\] done
clear
B)
\[KMn{{O}_{4}}\] done
clear
C)
\[KCl{{O}_{3}}\] done
clear
D)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
View Solution play_arrow
-
question_answer14)
Dry heating of calcium acetate gives [DPMT 1979, 81, 96; NCERT 1981; KCET 1993; Bihar CEE 1995; MNR 1986; MP PMT 1997; MP PET 1993, 95; JIPMER 2002; AIIMS 1996; CPMT 1982, 86, 96, 2003; RPMT 2002]
A)
Acetaldehyde done
clear
B)
Ethane done
clear
C)
Acetic acid done
clear
D)
Acetone done
clear
View Solution play_arrow
-
question_answer15)
Identify the product C in the series \[C{{H}_{3}}CN\xrightarrow{Na/{{C}_{2}}{{H}_{5}}OH}A\xrightarrow{HN{{O}_{2}}}B\xrightarrow{\text{Tollen }\!\!'\!\!\text{ s reagent}}C\] [MP PET 1999]
A)
\[C{{H}_{3}}COOH\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}NHOH\] done
clear
C)
\[C{{H}_{3}}CON{{H}_{2}}\] done
clear
D)
\[C{{H}_{3}}CHO\] done
clear
View Solution play_arrow
-
question_answer16)
Acetophenone is prepared by the reaction of which of the following in the presence of \[AlC{{l}_{3}}\] catalyst [AIIMS 1996]
A)
Phenol and acetic acid done
clear
B)
Benzene and acetone done
clear
C)
Benzene and acetyl chloride done
clear
D)
Phenol and acetone done
clear
View Solution play_arrow
-
question_answer17)
Isopropyl alcohol on oxidation gives [RPMT 1997; BHU 1997]
A)
Acetone done
clear
B)
Acetaldehyde done
clear
C)
Ether done
clear
D)
Ethylene done
clear
View Solution play_arrow
-
question_answer18)
On heating calcium acetate and calcium formate, the product formed is [DPMT 1984; EAMCET 1985; MP PMT 1996, 92; KCET 1990; CPMT 1979, 82, 84; BIT 1992; RPET 2000]
A)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}CHO\] done
clear
C)
\[HCHO+CaC{{O}_{3}}\] done
clear
D)
\[C{{H}_{3}}CHO+CaC{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer19)
Which of the following compound gives a ketone with Grignard reagent [CPMT 1988; MP PET 1997]
A)
Formaldehyde done
clear
B)
Ethyl alcohol done
clear
C)
Methyl cyanide done
clear
D)
Methyl iodide done
clear
View Solution play_arrow
-
question_answer20)
In the Rosenmund's reduction, \[BaS{{O}_{4}}\] taken with catalyst Pd acts as
A)
Promotor done
clear
B)
Catalytic poison done
clear
C)
Cooperator done
clear
D)
Absorber done
clear
View Solution play_arrow
-
question_answer21)
The Clemmenson reduction of acetone yields
A)
Ethanol done
clear
B)
Ethanal done
clear
C)
Propane done
clear
D)
Propanol done
clear
View Solution play_arrow
-
question_answer22)
Catalyst \[SnC{{l}_{2}}/HCl\] is used in [BHU 1995]
A)
Stephen's reduction done
clear
B)
Cannizzaro reaction done
clear
C)
Clemmensen's reduction done
clear
D)
Rosenmund's reduction done
clear
View Solution play_arrow
-
question_answer23)
Methyl ethyl ketone is prepared by the oxidation of [IIT-JEE 1987; MP PMT 1992]
A)
2-propanol done
clear
B)
1-butanol done
clear
C)
2-butanol done
clear
D)
t-butyl alcohol done
clear
View Solution play_arrow
-
question_answer24)
Benzaldehyde can be prepared by oxidation of toluene by [BHU 1986]
A)
Acidic \[KMn{{O}_{4}}\] done
clear
B)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
C)
\[Cr{{O}_{2}}C{{l}_{2}}\] done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer25)
\[{{C}_{6}}{{H}_{6}}+CO+HCl\xrightarrow{\text{Anhy }AlC{{l}_{3}}}X+HCl\] Compound X is [DPMT 1979, 83]
A)
\[{{C}_{6}}{{H}_{5}}C{{H}_{3}}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}Cl\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}CHO\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}COOH\] done
clear
View Solution play_arrow
-
question_answer26)
Which of the following gases when passed through warm dilute solution of \[{{H}_{2}}S{{O}_{4}}\] in presence of \[HgS{{O}_{4}}\] gives acetaldehyde [EAMCET 1986]
A)
\[C{{H}_{4}}\] done
clear
B)
\[{{C}_{2}}{{H}_{6}}\] done
clear
C)
\[{{C}_{2}}{{H}_{4}}\] done
clear
D)
\[{{C}_{2}}{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer27)
\[C{{H}_{3}}COC{{H}_{_{3}}}\] can be obtained by [CBSE PMT 1992]
A)
Heating acetaldehyde with methanol done
clear
B)
Oxidation of propyl alcohol done
clear
C)
Oxidation of isopropyl alcohol done
clear
D)
Reduction of propionic acid done
clear
View Solution play_arrow
-
question_answer28)
Propyne on hydrolysis in presence of HCl and \[HgS{{O}_{4}}\] gives [DPMT 1980; CPMT 1983]
A)
Acetaldehyde done
clear
B)
Acetone done
clear
C)
Formaldehyde done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer29)
Which of the following on reaction with \[N{{H}_{3}}\] gives urinary antiseptic compound [MP PMT 1999]
A)
HCHO done
clear
B)
\[C{{H}_{3}}CHO\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}CHO\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}CHO\] done
clear
View Solution play_arrow
-
question_answer30)
The oxidation product of 2-propanol with hot conc. \[HN{{O}_{3}}\] is [JIPMER 1997]
A)
Ethanoic acid done
clear
B)
Propanone done
clear
C)
Propanal done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer31)
Hydrolysis of ozonide of 1-butene gives [Kerala PMT 2003]
A)
Ethylene only done
clear
B)
Acetaldehyde and Formaldehyde done
clear
C)
Propionaldehyde and Formaldehyde done
clear
D)
Acetaldehyde only done
clear
E)
Acetaldehyde and Oxalic acid done
clear
View Solution play_arrow
-
question_answer32)
Ketones are prepared by
A)
Clemmensen's reduction done
clear
B)
Cannizzaro reaction done
clear
C)
Rosenmund's reduction done
clear
D)
Oppenaur's oxidation done
clear
View Solution play_arrow
-
question_answer33)
\[{{O}_{3}}\] reacts with \[C{{H}_{2}}=C{{H}_{2}}\] to form ozonide. On hydrolysis it forms [MP PET 1986, 90]
A)
Ethylene oxide done
clear
B)
HCHO done
clear
C)
Ethylene glycol done
clear
D)
Ethyl alcohol done
clear
View Solution play_arrow
-
question_answer34)
Ethyne on reaction with water in the presence of \[HgS{{O}_{4}}\]and \[{{H}_{2}}S{{O}_{4}}\] gives [UPSEAT 1999; BVP 2003]
A)
Acetone done
clear
B)
Acetaldehyde done
clear
C)
Acetic acid done
clear
D)
Ethyl alcohol done
clear
View Solution play_arrow
-
question_answer35)
\[C{{H}_{3}}-C{{H}_{2}}-C\equiv CH\underset{{{H}_{2}}S{{O}_{4}}}{\mathop{\xrightarrow{HgS{{O}_{4}}}}}\,\] A, the compound A is [Orissa JEE 2004]
A)
\[\overset{\overset{\,\,\,\,\,\,\,\,\,\,\,\,O}{\mathop{\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,||}}\,}{\mathop{C{{H}_{3}}-C{{H}_{2}}-C-C{{H}_{3}}}}\,\] done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-CHO\] done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-COOH\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer36)
When a mixture of methane and oxygen is passed through heated molybdenum oxide, the main product formed is [KCET 2004]
A)
Methanoic acid done
clear
B)
Ethanal done
clear
C)
Methanol done
clear
D)
Methanal done
clear
View Solution play_arrow
-
question_answer37)
Benzoin is [KCET 2004]
A)
Compound containing an aldehyde and a ketonic group done
clear
B)
a, b-unsaturated acid done
clear
C)
a-hydroxy aldehyde done
clear
D)
a-hydroxy ketone done
clear
View Solution play_arrow
-
question_answer38)
The oxidation of benzyl chloride with lead nitrate gives [MP PMT 2004]
A)
Benzyl alcohol done
clear
B)
Benzoic acid done
clear
C)
Benzaldehyde done
clear
D)
p-chlorobenzaldehyde done
clear
View Solution play_arrow
-
question_answer39)
\[R-CH=C{{H}_{2}}+CO+{{H}_{2}}\] \[\underset{\text{High Pressure}}{\mathop{\xrightarrow{\text{High Temp}}}}\,RC{{H}_{2}}C{{H}_{2}}CHO.\] [DPMT 2004] The above reaction is
A)
Mendius reaction done
clear
B)
Oxo process done
clear
C)
Sandorn's reaction done
clear
D)
Stephen's reaction done
clear
View Solution play_arrow
-
question_answer40)
Glycerol reacts with potassium bisulphate to produce [Pb. CET 2003]
A)
Allyl iodide done
clear
B)
Allyl sulphate done
clear
C)
Acryl aldehyde done
clear
D)
Glycerol trisulphate done
clear
View Solution play_arrow
-
question_answer41)
The reagent used in Gatterman Koch aldehyde synthesis is [CPMT 2004]
A)
\[Pb/BaS{{O}_{4}}\] done
clear
B)
alkaline \[KMn{{O}_{4}}\] done
clear
C)
acidic \[KMn{{O}_{4}}\] done
clear
D)
\[CO+HCl\] done
clear
View Solution play_arrow
-
question_answer42)
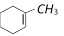
On reductive ozonolysis yields [Orissa JEE 2005]
A)
6-oxoheptanal done
clear
B)
6-oxoheptanoic acid done
clear
C)
6-hydroxyheptanal done
clear
D)
3-hydroxypentanal done
clear
View Solution play_arrow
-
question_answer43)
An alkene of molecular formula \[{{C}_{9}}{{H}_{18}}\] on ozonolysis gives 2, 2 dimethyl propanal & 2-butanon, then the alkene is [Kerala CET 2005]
A)
2, 2, 4-trimethyl ?3-hexene done
clear
B)
2, 2, 6-trimethyl-3-hexene done
clear
C)
2, 3, 4-trimethyl-2-hexene done
clear
D)
2, 2, 4-trimethyl-2-hexene done
clear
E)
2, 2dimethyl-2-heptene done
clear
View Solution play_arrow
![]() On reductive ozonolysis yields [Orissa JEE 2005]
On reductive ozonolysis yields [Orissa JEE 2005] 