-
question_answer1)
In fructose, the possible optical isomers are
A)
12 done
clear
B)
8 done
clear
C)
16 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer2)
Glucose molecule reacts with X number of molecules of phenylhydrazine to yield osazone. The value of X is
A)
three done
clear
B)
two done
clear
C)
one done
clear
D)
four done
clear
View Solution play_arrow
-
question_answer3)
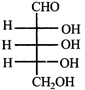
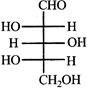
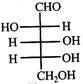
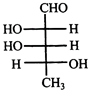
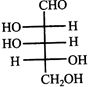
Which L-sugar on oxidation gives an optically active dibasic acid (2 COOH groups)?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer4)
In cells the net production of ATP molecules generated from one glucose molecule is
A)
46 done
clear
B)
32 done
clear
C)
36 done
clear
D)
40 done
clear
View Solution play_arrow
-
question_answer5)
Which is correct statement?
A)
Starch is a polymer of \[\alpha \]-glucose done
clear
B)
In cyclic structure of fructose, there are four carbons and one oxygen atom done
clear
C)
Amylose is a component of cellulose done
clear
D)
Proteins are composed of only one type of amino acids done
clear
View Solution play_arrow
-
question_answer6)
Vitamin D is also known as
A)
sunshine vitamin done
clear
B)
ascorbic acid done
clear
C)
growth vitamin done
clear
D)
reproductive vitamin done
clear
View Solution play_arrow
-
question_answer7)
Night blindness is caused by deficiency of
A)
vitamin \[{{B}_{12}}\] done
clear
B)
vitamin A done
clear
C)
vitamin C done
clear
D)
vitamin E done
clear
View Solution play_arrow
-
question_answer8)
The hormone which controls the processes of burning of fats, proteins and carbohydrates to liberate energy in the body is
A)
cortisone done
clear
B)
adrenaline done
clear
C)
thyroxine done
clear
D)
insulin done
clear
View Solution play_arrow
-
question_answer9)
Two vitamins absorbed from intestine along with fast are
A)
A, D done
clear
B)
A, B done
clear
C)
A, C done
clear
D)
D, B done
clear
View Solution play_arrow
-
question_answer10)
Which one of the following is an amine hormone?
A)
Thyroxine done
clear
B)
Oxypurin done
clear
C)
Insulin done
clear
D)
Progesterone done
clear
View Solution play_arrow
-
question_answer11)
Complete hydrolysis of starch gives:
A)
glucose only done
clear
B)
galactose and fructose in equimolar amounts done
clear
C)
glucose and galactose in equimolar amounts done
clear
D)
glucose and fructose in equimolar amounts done
clear
View Solution play_arrow
-
question_answer12)
Accumulation of which of the following molecules in the muscles occurs as a result of vigorous exercise?
A)
Glycogen done
clear
B)
Glucose done
clear
C)
Pyruvic acid done
clear
D)
L-lactic acid done
clear
View Solution play_arrow
-
question_answer13)
Which of the following will not show mutarotation?
A)
Maltose done
clear
B)
Lactose done
clear
C)
Glucose done
clear
D)
Sucrose done
clear
View Solution play_arrow
-
question_answer14)
An organic compound with the formula \[{{C}_{6}}{{H}_{12}}{{O}_{6}}\]forms a yellow crystalline solid with phenylhydrazine and gives a mixture of sorbitol and mannitol when reduced with sodium. Which among the following could be the compound?
A)
fructose done
clear
B)
glucose done
clear
C)
mannose done
clear
D)
sucrose done
clear
View Solution play_arrow
-
question_answer15)
When \[\alpha \]-D-glucose and \[\beta \]-D-glucose are dissolved in water in two separate beakers I and II respectively and allowed to stand, then -
A)
specific rotation in beaker I will decrease while in II will increase upto a constant value done
clear
B)
the specific rotation of equilibrium mixture in two beakers will be different done
clear
C)
the equilibrium mixture in both beakers will be leavorotatory done
clear
D)
the equilibrium mixture in both beakers will contain only cyclic form of glucose done
clear
View Solution play_arrow
-
question_answer16)
The \[p{{K}_{4}}\] values for the three ionizable groups X, Y and Z of glutamic acid are 4.3, 9.7 and 2.2 respectively \[\underset{X}{\mathop{H{{O}_{2}}C}}\,-C{{H}_{2}}-\underset{Y}{\mathop{\underset{_{+}N{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,H}}\,-\underset{Z}{\mathop{C{{O}_{2}}H}}\,\] The isoelectric point for the amino acid is:
A)
7.00 done
clear
B)
3.25 done
clear
C)
4.95 done
clear
D)
5.95 done
clear
View Solution play_arrow
-
question_answer17)
A vitamin that contains both N and P is
A)
Vitamin C done
clear
B)
Vitamin K done
clear
C)
Vitamin \[{{B}_{12}}\] done
clear
D)
Vitamin D done
clear
View Solution play_arrow
-
question_answer18)
Match List I (name of vitamin) with List II (deficiency result/disease) and select the correct answer using the codes given below the lists:
| List I | List II |
| I. Ascorbic acid | A. Beri-beri |
| II. Retinol | B. Cracked lips |
| III. Riboflavin | C. Scurvy |
| IV. Thiamine | D. Night blindness |
A)
I-B, II-A, III-C, IV-D done
clear
B)
I-A, II-B, III-C, IV-D done
clear
C)
I-D, II-C, III-B, IV-A done
clear
D)
I-C, II-D, III-B, IV-A done
clear
View Solution play_arrow
-
question_answer19)
Nucleotides in DNA are linked by -
A)
hydrogen bond done
clear
B)
3', 5'-phosphodiester bond done
clear
C)
glycosidic bond done
clear
D)
peptide bond done
clear
View Solution play_arrow
-
question_answer20)
If one strand of DNA has the sequence ATGCTTGA, the sequence in the complimentary strand would be
A)
TACGAACT done
clear
B)
TCCGAACT done
clear
C)
TACGTACT done
clear
D)
TACGTAGT done
clear
View Solution play_arrow
-
question_answer21)
Which of the following pairs can be distinguished by Fehling's solution?
A)
Glucose and fructose done
clear
B)
Glucose and sucrose done
clear
C)
Methanal and ethanol done
clear
D)
Hydroxypropanone and benzaldehyde. done
clear
View Solution play_arrow
-
question_answer22)
Which of the following statement(s) is (are) true?
| (i) All amino acids contain one chiral center. |
| (ii) Some amino acids contain one, while some contain more chiral center or even no chiral center. |
| (iii) All amino acids found in proteins have L configuration. |
| (iv) All amino acids found in proteins have \[1{}^\circ \] amino group. |
A)
(ii), (iii) & (iv) done
clear
B)
(ii) & (iii) done
clear
C)
(i), (iii) & (iv) done
clear
D)
(i) & (iv). done
clear
View Solution play_arrow
-
question_answer23)
In osazone formation, glucose reacts with three molecules of phenylhydrazine. Which statement is true regarding this?
A)
All the three molecules react in similar fashion done
clear
B)
Two molecules react in similar manner, while the third reacts in different way done
clear
C)
All the three molecules react in different ways done
clear
D)
None of the above is true done
clear
View Solution play_arrow
-
question_answer24)
Natural glucose is termed D-glucose because:
A)
\[-OH\] on the second carbon is on the right side in Fischer projection done
clear
B)
\[-OH\] on the sixth carbon is on the right side in Fischer projection. done
clear
C)
\[-OH\] on the fifth carbon is on the right side in Fischer projection. done
clear
D)
It is dextrorotatory. done
clear
View Solution play_arrow
-
question_answer25)
Which of the following statement is not correct?
A)
Amylopectin is a branched polymer of \[\alpha \]- glucose. done
clear
B)
Cellulose is a linear polymer of \[\beta \]-glucose done
clear
C)
Glycogen is the food reserve of plants done
clear
D)
All proteins are polymers of \[\alpha \]- amino acids. done
clear
View Solution play_arrow
-
question_answer26)
Amylopectin is a polymer of
A)
\[\alpha -D\]-glucose done
clear
B)
amino acid done
clear
C)
\[\beta -D\]- glucose done
clear
D)
amylase. done
clear
View Solution play_arrow
-
question_answer27)
Which of the following is a non-reducing sugar?
A)
Lactose done
clear
B)
Fructose done
clear
C)
Sucrose done
clear
D)
Maltose done
clear
View Solution play_arrow
-
question_answer28)
Glycosidic linkage is actually an:
A)
Carbonyl bond done
clear
B)
Ether bond done
clear
C)
Ester bond done
clear
D)
Amide bond done
clear
View Solution play_arrow
-
question_answer29)
Which of the following does not reduce Benedict's solution?
A)
Glucose done
clear
B)
Fructose done
clear
C)
Sucrose done
clear
D)
Aldehyde done
clear
View Solution play_arrow
-
question_answer30)
Hydrolysis of sucrose is called
A)
hydration done
clear
B)
saponification done
clear
C)
esterification done
clear
D)
inversion done
clear
View Solution play_arrow
-
question_answer31)
Assume that a particular amino acid has an isoelectric point of 6.0. In a solution at pH 1.0, which of the following species will predominate?
A)
\[{{H}_{3}}\overset{+}{\mathop{N}}\,\overset{R}{\mathop{\overset{|}{\mathop{C}}\,}}\,HC{{O}_{2}}H\] done
clear
B)
\[{{H}_{2}}\overset{{}}{\mathop{N}}\,\overset{R}{\mathop{\overset{|}{\mathop{C}}\,}}\,HC{{O}_{2}}H\] done
clear
C)
\[{{H}_{3}}\overset{+}{\mathop{N}}\,\overset{R}{\mathop{\overset{|}{\mathop{C}}\,}}\,HC{{O}_{2}}\] done
clear
D)
\[{{H}_{2}}\overset{{}}{\mathop{N}}\,\overset{R}{\mathop{\overset{|}{\mathop{C}}\,}}\,HCO_{2}^{-}\] done
clear
View Solution play_arrow
-
question_answer32)
A mixture of two amino acids having pI 9.60 and 5.40 can be separated
A)
by adjusting the pH of the solution at 9.60 done
clear
B)
by adjusting the pH of the solution at 4.20 done
clear
C)
by adjusting the pH of the solution at 7.0 done
clear
D)
by adjusting the pH of the solution at 7.5. done
clear
View Solution play_arrow
-
question_answer33)
The correct statement in respect of protein haemoglobin is that it
A)
acts as an oxygen carrier in the blood done
clear
B)
forms antibodies and offers resistance to diseases done
clear
C)
enzymes are specific biological catalysts that can normally function at very high temperature \[\left( T\tilde{\ }1000K \right)\] done
clear
D)
enzymes are specific biological catalysts that possess well-defined active sites done
clear
View Solution play_arrow
-
question_answer34)
Which of the following carbohydrates is not related to (+)-glucose?
A)
Amylopectin done
clear
B)
Amylose done
clear
C)
Inulin done
clear
D)
Glycogen done
clear
View Solution play_arrow
-
question_answer35)
What will happen when D-(+)-glucose is treated with methanolic \[-HCl\] followed by Tollens' reagent?
A)
A black ppt. will be formed done
clear
B)
A red ppt. will be formed done
clear
C)
A green colour will appear done
clear
D)
No characteristic colour or ppt. will be formed. done
clear
View Solution play_arrow
-
question_answer36)
Glucose-D has a great tendency to be converted into cyclic isomer. Which two carbon atoms get joined through 'O' to form this hemiacetal?
A)
\[{{C}_{1}}\] and \[{{C}_{4}}\] done
clear
B)
\[{{C}_{1}}\]and \[{{C}_{5}}\] done
clear
C)
\[{{C}_{1}}\] and \[{{C}_{6}}\] done
clear
D)
\[{{C}_{2}}\] and \[{{C}_{6}}\] done
clear
View Solution play_arrow
-
question_answer37)
Among the three compounds shown below, two yield the same product on reaction with warm\[HN{{O}_{3}}\]. The exception is:
A)
B)
C)
D)
None of these done
clear
View Solution play_arrow
-
question_answer38)
The enantiomer of \[\alpha -D-\left( + \right)\]-glucose is
A)
\[\beta -D-\left( + \right)\]-glucose done
clear
B)
\[\alpha -D-\left( - \right)\] -glucose done
clear
C)
\[\alpha -L-\left( - \right)\]-glucose done
clear
D)
\[\beta -L-\left( - \right)\]-glucose. done
clear
View Solution play_arrow
-
question_answer39)
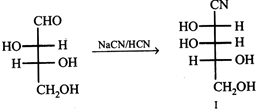 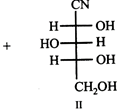 |
| Compounds I and II may be grouped as |
A)
diastereomers done
clear
B)
epimers done
clear
C)
C-2 epimers done
clear
D)
all of the three. done
clear
View Solution play_arrow
-
question_answer40)
Which of the following evolves carbon dioxide, on oxidation with periodate?
A)
B)
C)
Both done
clear
D)
None done
clear
View Solution play_arrow
-
question_answer41)
Imino acid among these compounds is
A)
serine done
clear
B)
proline done
clear
C)
tyrosine done
clear
D)
lysine done
clear
View Solution play_arrow
-
question_answer42)
Which amino acid is achiral?
A)
alanine done
clear
B)
valine done
clear
C)
proline done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer43)
Which is not a true statement?
A)
\[\alpha \]-carbon of \[\alpha \]-amino acid is asymmetric done
clear
B)
All proteins are found in L-form done
clear
C)
Human body can synthesize all proteins they need done
clear
D)
At pH = 7 both amino acids and carboxylic groups exist in the ionised form done
clear
View Solution play_arrow
-
question_answer44)
Which of the following is a steroid hormone?
A)
Cholesterol done
clear
B)
Adrenaline done
clear
C)
Thyroxine done
clear
D)
Progesterone done
clear
View Solution play_arrow
-
question_answer45)
Vitamin \[{{B}_{6}}\] is known as
A)
pyridoxine done
clear
B)
thiamine done
clear
C)
tocopherol done
clear
D)
riboflavin done
clear
View Solution play_arrow
-
question_answer46)
A sequence of how many nucleotides in messenger RNA makes a codon for an amino acid?
A)
Three done
clear
B)
Four done
clear
C)
One done
clear
D)
Two done
clear
View Solution play_arrow
-
question_answer47)
The process by which synthesis of protein takes place based on the genetic information present in m-RNA is called
A)
Translation done
clear
B)
Transcription done
clear
C)
Replication done
clear
D)
Messenger hypothesis done
clear
View Solution play_arrow
-
question_answer48)
Which of the following substances is not present in nucleic acids?
A)
Cytosine done
clear
B)
Adenine done
clear
C)
Thymine done
clear
D)
Guanidine done
clear
View Solution play_arrow
-
question_answer49)
Which of the following is not present in a nucleotide?
A)
Guanine done
clear
B)
Cytosine done
clear
C)
Adenine done
clear
D)
Tyrosine done
clear
View Solution play_arrow
-
question_answer50)
Iso-electric point of alanine is (pH = 6). At which pH, maximum concentration of zwitter ion of alanine will be present?
A)
pH>6 done
clear
B)
pH<6 done
clear
C)
pH=6 done
clear
D)
pH=7 done
clear
View Solution play_arrow
-
question_answer51)
\[H-C\equiv C-H\xrightarrow[{{H}_{2}}S{{O}_{4}}]{HgS{{O}_{4}}}(A)\] \[\xrightarrow[(2)\,{{H}_{3}}{{O}^{+}}]{(1)\,N{{H}_{3}}+HCN}(B);\] Product [B] of given reaction is:
A)
Glycine done
clear
B)
Alanine done
clear
C)
valine done
clear
D)
Leucine done
clear
View Solution play_arrow
-
question_answer52)
The dipeptide, Gly. Ala has structure -
A)
\[{{H}_{3}}\overset{+}{\mathop{N}}\,C{{H}_{2}}\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,NH\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,HC{{\bar{O}}_{2}}\] done
clear
B)
\[{{H}_{3}}\overset{+}{\mathop{N}}\,\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,H\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,NH\underset{{}}{\mathop{\underset{{}}{\mathop{C}}\,}}\,{{H}_{2}}C{{\overset{-}{\mathop{O}}\,}_{2}}\] done
clear
C)
\[{{\overset{-}{\mathop{O}}\,}_{2}}C\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,HNH-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-\underset{{}}{\mathop{\underset{{}}{\mathop{C}}\,}}\,{{H}_{2}}\overset{+}{\mathop{N}}\,{{H}_{3}}\] done
clear
D)
\[{{\overset{-}{\mathop{O}}\,}_{2}}CC{{H}_{2}}NH-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,H\overset{+}{\mathop{N}}\,{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer53)
All of the following statements apply to proteins except
A)
Proteins generally have no definite melting point done
clear
B)
Proteins contain the grouping -CONH- done
clear
C)
Proteins have high molecular weight done
clear
D)
Proteins can only contain the elements C, H, O and N. done
clear
View Solution play_arrow
-
question_answer54)
The reason for double helical structure of DNA is the operation of:
A)
Electrostatic attractions done
clear
B)
van der Waals forces done
clear
C)
Dipole - Dipole interactions done
clear
D)
Hydrogen bonding done
clear
View Solution play_arrow
-
question_answer55)
Among the following organic acids, the acid present in rancid butter is:
A)
Pyruvicacid done
clear
B)
Lactic acid done
clear
C)
Butyric acid done
clear
D)
Acetic acid done
clear
View Solution play_arrow
-
question_answer56)
Which of the following enzyme converts starch into maltose?
A)
Diastase done
clear
B)
Maltase done
clear
C)
Zymase done
clear
D)
Invertase done
clear
View Solution play_arrow
-
question_answer57)
Which statement is incorrect about peptide bond?
A)
C-N bond length in proteins is longer than usual C-N bond length done
clear
B)
Spectroscopic analysis shows planar structure of \[-\underset{O}{\mathop{\underset{||}{\mathop{C}}\,}}\,-NH-\] bond done
clear
C)
C-N bond length in proteins is smaller than usual C-N bond length done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer58)
\[D-\left( + \right)-Glucose~\xrightarrow{5{{\left( C{{H}_{3}}CO \right)}_{2}}O}D-\left( + \right)-Glucose\] pentaacetate Which statement is true about glucose pentaacetate?
A)
It will react with phenylhydrazine but not with Tollens' reagent. done
clear
B)
It will react with Tollens' reagent but not with phenylhydrazine. done
clear
C)
It will react with both of the above mentioned reagents. done
clear
D)
It will react neither with phenylhydrazine nor with Tollens' reagent. done
clear
View Solution play_arrow
-
question_answer59)
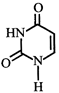
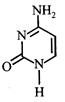
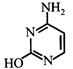
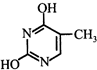
Which of the following structures represents thymine?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer60)
Among the following vitamins the one whose deficiency causes rickets (bone deficiency) is
A)
Vitamin A done
clear
B)
Vitamin B done
clear
C)
Vitamin D done
clear
D)
Vitamin C done
clear
View Solution play_arrow
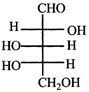 done
clear
done
clear
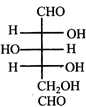 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear


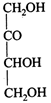 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
