-
question_answer1)
Ethyl ester\[\underset{\text{excess}}{\mathop{\xrightarrow{C{{H}_{3}}MgBr}}}\,P\]. The product P will be [IIT-JEE 2003]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer2)
Hydrogenation of \[{{C}_{6}}{{H}_{5}}CHOH-COOH\] over \[Rh-A{{l}_{2}}{{O}_{3}}\] catalyst in methanol gives [Roorkee Qualifying 1998]
A)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}COOH\] done
clear
B)
\[{{C}_{6}}{{H}_{11}}CHOHCOOH\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}CHOHC{{H}_{2}}OH\] done
clear
D)
\[{{C}_{6}}{{H}_{11}}C{{H}_{2}}COOH\] done
clear
View Solution play_arrow
-
question_answer3)
Which of the following has the most acidic proton [Roorkee Qualifying 1998]
A)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
B)
\[{{(C{{H}_{3}})}_{2}}C=C{{H}_{2}}\] done
clear
C)
\[C{{H}_{3}}COC{{H}_{2}}COC{{H}_{3}}\] done
clear
D)
\[{{(C{{H}_{3}}CO)}_{3}}CH\] done
clear
View Solution play_arrow
-
question_answer4)
In the anion \[HCO{{O}^{-}}\] the two carbon-oxygen bonds are found to be of equal length. What is the reason for it
A)
Electronic orbitals of carbon atom are hybridised done
clear
B)
The C = O bond is weaker than the C - O bond done
clear
C)
The anion \[HCO{{O}^{-}}\] has two resonating structures done
clear
D)
The anion is obtained by removal of a proton form the acid molecule done
clear
View Solution play_arrow
-
question_answer5)
An organic compound of molecular formula \[{{C}_{4}}{{H}_{10}}O\] does not react with sodium. With excess of \[HI,\] it gives only one type of alkyl halide. The compound is [SCRA 2001]
A)
Ethoxyethane done
clear
B)
2-Methoxypropane done
clear
C)
1-Methoxypropane done
clear
D)
1-Butanol done
clear
View Solution play_arrow
-
question_answer6)
When \[C{{H}_{2}}=CH-COOH\] is reduced with \[LiAl{{H}_{4}}\], the compound obtained will be [AIEEE 2003]
A)
\[C{{H}_{3}}-C{{H}_{2}}-COOH\] done
clear
B)
\[C{{H}_{2}}=CH-C{{H}_{2}}OH\] done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}OH\] done
clear
D)
\[C{{H}_{3}}-C{{H}_{2}}-CHO\] done
clear
View Solution play_arrow
-
question_answer7)
In a set of the given reactions, acetic acid yielded a product C \[C{{H}_{3}}COOH+PC{{l}_{5}}\to A\underset{anh.\,AlC{{l}_{3}}}{\mathop{\xrightarrow{{{C}_{6}}{{H}_{6}}}}}\,\]\[B\underset{ether}{\mathop{\xrightarrow{{{C}_{2}}{{H}_{5}}MgBr}}}\,C\].Product C would be [CBSE PMT 2003]
A)
\[\overset{\overset{{{C}_{2}}{{H}_{5}}}{\mathop{|\,\,\,\,\,\,\,\,\,\,}}\,}{\mathop{C{{H}_{3}}-C(OH){{C}_{6}}{{H}_{5}}}}\,\] done
clear
B)
\[C{{H}_{3}}CH(OH){{C}_{2}}{{H}_{5}}\] done
clear
C)
\[C{{H}_{3}}CO{{C}_{6}}{{H}_{5}}\] done
clear
D)
\[C{{H}_{3}}CH(OH){{C}_{6}}{{H}_{5}}\] done
clear
View Solution play_arrow
-
question_answer8)
Carboxylic acids are more acidic than phenol and alcohol because of [Tamil Nadu CET 2001]
A)
Intermolecular hydrogen bonding done
clear
B)
Formation of dimers done
clear
C)
Highly acidic hydrogen done
clear
D)
Resonance stabilization of their conjugate base done
clear
View Solution play_arrow
-
question_answer9)
\[R-C{{H}_{2}}-C{{H}_{2}}OH\] can be converted into \[RC{{H}_{2}}C{{H}_{2}}COOH.\] The correct sequence of the reagents is [AIIMS 1997]
A)
\[PB{{r}_{3}},KCN,{{H}_{3}}{{O}^{+}}\] done
clear
B)
\[PB{{r}_{3}},KCN,{{H}_{2}}\] done
clear
C)
\[HCN,PB{{r}_{3}},{{H}^{+}}\] done
clear
D)
\[KCN,{{H}^{+}}\] done
clear
View Solution play_arrow
-
question_answer10)
When propionic acid is treated with aqueous sodium bicarbonate \[C{{O}_{2}}\] is liberated. The 'C' of \[C{{O}_{2}}\] comes from [IIT-JEE (Screening) 1999]
A)
Methyl group done
clear
B)
Carboxylic acid group done
clear
C)
Methylene group done
clear
D)
Bicarbonate done
clear
View Solution play_arrow
-
question_answer11)
Benzoyl chloride is prepared from benzoic acid by [IIT-JEE (Screening) 2000]
A)
\[C{{l}_{2}},\,\,hv\] done
clear
B)
\[S{{O}_{2}}C{{l}_{2}}\] done
clear
C)
\[SOC{{l}_{2}}\] done
clear
D)
\[C{{l}_{2}},{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer12)
Identify the correct order of boiling points of the following compounds \[\underset{(1)}{\mathop{C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OH}}\,\],\[\underset{(2)}{\mathop{C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}CHO}}\,\], \[\underset{(3)}{\mathop{C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}COOH}}\,\] [IIT-JEE (Screening) 2002]
A)
\[1>2>3\] done
clear
B)
\[3>1>2\] done
clear
C)
\[1>3>2\] done
clear
D)
\[3>2>1\] done
clear
View Solution play_arrow
-
question_answer13)
The compound not soluble in acetic acid is [UPSEAT 2003; IIT-JEE 1986]
A)
\[CaC{{O}_{3}}\] done
clear
B)
\[CaO\] done
clear
C)
\[Ca{{C}_{2}}{{O}_{4}}\] done
clear
D)
\[Ca{{(OH)}_{2}}\] done
clear
View Solution play_arrow
-
question_answer14)
The ortho/para directing group among the following is [AIIMS 2003]
A)
\[COOH\] done
clear
B)
\[CN\] done
clear
C)
\[COC{{H}_{3}}\] done
clear
D)
\[NHCOC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer15)
Iodoform test is not given by [BHU 1995]
A)
Acetone done
clear
B)
Ethyl alcohol done
clear
C)
Acetic acid done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer16)
How will you convert butan-2-one to propanoic acid [IIT-JEE (Screening) 2005]
A)
Tollen's reagent done
clear
B)
Fehling's solution done
clear
C)
\[NaOH/{{I}_{2}}/{{H}^{+}}\] done
clear
D)
\[NaOH/NaI/{{H}^{+}}\] done
clear
View Solution play_arrow
-
question_answer17)
Which of the acids cannot be prepared by Grignard reagent [MH CET 2004]
A)
Acetic acid done
clear
B)
Succinic acid done
clear
C)
Formic acid done
clear
D)
All of these done
clear
View Solution play_arrow
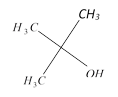 done
clear
done
clear
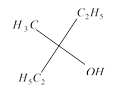 done
clear
done
clear
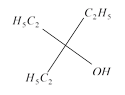 done
clear
done
clear
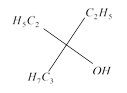 done
clear
done
clear
