-
question_answer1)
The first law of thermodynamics is only
A)
The law of conservation of energy done
clear
B)
The law of conservation of mass done
clear
C)
The law of conservation of momentum done
clear
D)
Both A and B done
clear
View Solution play_arrow
-
question_answer2)
A mixture of two moles of carbon monoxide and one mole of oxygen, in a closed vessel is ignited to convert the carbon monoxide to carbon dioxide. If \[\Delta H\] is the enthalpy change and \[\Delta E\] is the change in internal energy, then [KCET 2005]
A)
\[\Delta H>\Delta E\] done
clear
B)
\[\Delta H<\Delta E\] done
clear
C)
\[\Delta H=\Delta E\] done
clear
D)
The relationship depends on the capacity of the vessel done
clear
View Solution play_arrow
-
question_answer3)
Which of the following is always negative for exothermic reaction? [BCECE 2005]
A)
DH done
clear
B)
DS done
clear
C)
DG done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer4)
The relation between \[\Delta E\] and \[\Delta H\] is [MP PET 1992; MP PMT 1996; MP PET/PMT 1998]
A)
\[\Delta H=\Delta E-P\,\Delta V\] done
clear
B)
\[\Delta H=\Delta E+P\,\Delta V\] done
clear
C)
\[\Delta E=\Delta V+\Delta H\] done
clear
D)
\[\Delta E=\Delta H+P\,\Delta V\] done
clear
View Solution play_arrow
-
question_answer5)
At constant T and P, which one of the following statements is correct for the reaction, \[CO(g)+\frac{1}{2}{{O}_{2}}(g)\to C{{O}_{2}}(g)\] [AIIMS 1982, 83; KCET 1988; BHU 1995; MP PET 1997, 99]
A)
\[\Delta H\] is independent of the physical state of the reactants of that compound done
clear
B)
\[\Delta H\,>\,\Delta E\] done
clear
C)
\[\Delta H\,<\,\Delta E\] done
clear
D)
\[\Delta H=\Delta E\] done
clear
View Solution play_arrow
-
question_answer6)
For the reaction of one mole of zinc dust with one mole of sulphuric acid in a bomb calorimeter, \[\Delta \text{U}\] and w correspond to [AIIMS 2005]
A)
\[\Delta \text{U}<0,\ w=0\] done
clear
B)
\[\Delta \text{U}=0,\ w<0\] done
clear
C)
\[\Delta \text{U}>0,\ w=0\] done
clear
D)
\[\Delta \text{U}<0,\ w>0\] done
clear
View Solution play_arrow
-
question_answer7)
Which is not the correct relation between enthalpy \[(\Delta H)\]and intrinsic energy \[(\Delta E)\] [EAMCET 1992]
A)
\[\Delta H=\Delta E+\,P\,\Delta V\] done
clear
B)
\[\Delta H\,=\,\Delta E\,+\,n\,RT\] done
clear
C)
\[\Delta H=\Delta E\,-\,P\,\Delta V\] done
clear
D)
\[\Delta E=\Delta H\,-\,P\,\Delta V\] done
clear
View Solution play_arrow
-
question_answer8)
The law of Lavoisier and Laplace illustrates [KCET 1989]
A)
The principle of conservation of energy done
clear
B)
Equivalence of mechanical and thermal energy done
clear
C)
The principle of conservation of matter done
clear
D)
Equivalence of mechanical and chemical energy done
clear
View Solution play_arrow
-
question_answer9)
For the reaction \[{{N}_{2}}+3{{H}_{2}}\]⇌\[2N{{H}_{3}};\,\Delta H=\] [Roorkee 2000; CBSE PMT 1991, 2002]
A)
\[\Delta E-RT\] done
clear
B)
\[\Delta E-2RT\] done
clear
C)
\[\Delta E+RT\] done
clear
D)
\[\Delta E+2RT\] done
clear
View Solution play_arrow
-
question_answer10)
If \[\Delta H\] is the change in enthalpy and \[\Delta E\] the change in internal energy accompanying a gaseous reaction KCET 1989; CBSE PMT 1990]
A)
\[\Delta H\] is always greater than \[\Delta E\] done
clear
B)
\[\Delta H<\Delta E\] only if the number of moles of the products is greater than the number of the reactants done
clear
C)
\[\Delta H\] is always less than \[\Delta E\] done
clear
D)
\[\Delta H<\Delta E\] only if the number of moles of the products is less than the number of moles of the reactants done
clear
View Solution play_arrow
-
question_answer11)
?The resultant heat change in a reaction is the same whether it takes place in one or several stages.? This statement is called [MP PMT/PET 1988; MP PMT 1989]
A)
Lavoisier and Laplace law done
clear
B)
Hess's law done
clear
C)
Joule's law done
clear
D)
Le?chatelier's principle done
clear
View Solution play_arrow
-
question_answer12)
Hess's law of constant heat summation in based on [MP PET 2001]
A)
\[E=m{{c}^{2}}\] done
clear
B)
Conservation of mass done
clear
C)
First law of thermodynamics done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer13)
Consider the reaction : \[{{N}_{2}}+3{{H}_{2}}\to 2N{{H}_{3}}\] carried out at constant temperature and pressure. If \[\Delta H\] and \[\Delta U\] are the enthalpy and internal energy changes for the reaction, which of the following expression is true [AIEEE 2005]
A)
\[\Delta H=0\] done
clear
B)
\[\Delta H=\Delta U\] done
clear
C)
\[\Delta H<\Delta U\] done
clear
D)
\[\Delta H>\Delta U\] done
clear
View Solution play_arrow
-
question_answer14)
Which of the following is the correct equation [CBSE PMT 1996]
A)
\[\Delta U=\Delta Q-W\] done
clear
B)
\[\Delta W=\Delta U+\Delta Q\] done
clear
C)
\[\Delta U=\Delta W+\Delta Q\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer15)
Hess law is applicable for the determination of heat of [AIIMS 1998; Pb. PET/PMT 1999]
A)
Reaction done
clear
B)
Formation done
clear
C)
Transition done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer16)
Enthalpy for the reaction \[C+{{O}_{2}}\to C{{O}_{2}}\] is [DPMT 1987, 91]
A)
Positive done
clear
B)
Negative done
clear
C)
Zero done
clear
D)
None done
clear
View Solution play_arrow
-
question_answer17)
During an isothermal expansion of an ideal gas its [CBSE PMT 1991]
A)
Internal energy increases done
clear
B)
Enthalpy decreases done
clear
C)
Enthalpy remains unaffected done
clear
D)
Enthalpy reduces to zero done
clear
View Solution play_arrow
-
question_answer18)
The work done in ergs for the reversible expansion of one mole of an ideal gas from a volume of 10 litres to 20 litres at \[{{25}^{o}}C\] is [CMC Vellore 1991]
A)
\[2.303\times 298\times 0.082\,\log \,2\] done
clear
B)
\[298\times {{10}^{7}}\times 8.31\times 2.303\,\log \,2\] done
clear
C)
\[2.303\times 298\times 0.082\,\log \,0.5\] done
clear
D)
\[8.31\times {{10}^{7}}\times 298-2.303\,\log \,0.5\] done
clear
E)
\[2.303\times 298\times 2\,\log \,2\] done
clear
View Solution play_arrow
-
question_answer19)
The law of conservation of energy states that [NCERT 1984]
A)
The internal energy of a system is constant done
clear
B)
The heat content of a system is constant done
clear
C)
Energy is neither created nor destroyed done
clear
D)
There is an equivalence between energy and mass done
clear
View Solution play_arrow
-
question_answer20)
The enthalpies of the elements in their standard states are assumed to be
A)
Zero at 298 K done
clear
B)
Unit at 298 K done
clear
C)
Zero at all temperatures done
clear
D)
Zero at 273 K done
clear
View Solution play_arrow
-
question_answer21)
The heat \[Q\] for a reaction at constant volume is equal to
A)
\[{{H}_{P}}-{{H}_{R}}\] done
clear
B)
\[{{H}_{R}}-{{H}_{P}}\] done
clear
C)
\[{{E}_{P}}-{{E}_{R}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer22)
Which of the following expressions represents the first law of thermodynamics [MP PET 1996, 2000; AFMC 1997; BHU 1999; AMU 2000; KCET (Med.) 2000, 01; CBSE PMT 2000; MP PMT 2002]
A)
\[\Delta E=-q+W\] done
clear
B)
\[\Delta E=q-W\] done
clear
C)
\[\Delta E=q+W\] done
clear
D)
\[\Delta E=-q-W\] done
clear
View Solution play_arrow
-
question_answer23)
In a reversible isothermal process, the change in internal energy is
A)
Zero done
clear
B)
Positive done
clear
C)
Negative done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer24)
A schematic plot of \[\ln {{K}_{eq}}\]versus inverse of temperature for a reaction is shown below
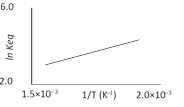
The reaction must be [AIEEE 2005]
A)
Exothermic done
clear
B)
Endothermic done
clear
C)
One with negligible enthalpy change done
clear
D)
Highly spontaneous at ordinary temperature done
clear
View Solution play_arrow
-
question_answer25)
Hess law of heat summation includes [AFMC 1992]
A)
Initial reactants only done
clear
B)
Initial reactants and final products done
clear
C)
Final products only done
clear
D)
Intermediates only done
clear
View Solution play_arrow
-
question_answer26)
An ideal gas at constant temperature and pressure expands, then its [BHU 1998]
A)
Internal energy remains same done
clear
B)
Internal energy decreases done
clear
C)
Internal energy increases done
clear
D)
Entropy first increases and then decreases done
clear
View Solution play_arrow
-
question_answer27)
The internal energy of a substance [KCET 1998; AFMC 2001; AIIMS 2001]
A)
Increases with increase in temperature done
clear
B)
Decreases with increase in temperature done
clear
C)
Can be calculated by the relation \[E=m{{c}^{2}}\] done
clear
D)
Remains unaffected with change in temperature done
clear
View Solution play_arrow
-
question_answer28)
The relation between change in internal energy \[(\Delta E)\], change in enthalpy \[(\Delta H)\] and work done (W) is represented as [AFMC 1999]
A)
\[\Delta H=\Delta E+W\] done
clear
B)
\[W=\Delta E-\Delta H\] done
clear
C)
\[\Delta E=W-\Delta H\] done
clear
D)
\[\Delta E=\Delta H+W\] done
clear
View Solution play_arrow
-
question_answer29)
The enthalpy of neutralization of which of the following acids and bases is nearly \[-13.6\,Kcal\] [Roorkee 1999]
A)
HCN and NaOH done
clear
B)
HCl and KOH done
clear
C)
HCl and NaOH done
clear
D)
HCl and NH4OH done
clear
View Solution play_arrow
-
question_answer30)
Work done during isothermal expansion of one mole of an ideal gas from 10 atm to 1 atm at 300 K is (Gas constant = 2) [AIIMS 2000]
A)
938.8 cal. done
clear
B)
1138.8 cal. done
clear
C)
1381.8 cal. done
clear
D)
1581.8 cal. done
clear
View Solution play_arrow
-
question_answer31)
Joule-Thomson expansion is [JIPMER 2000]
A)
Isobaric done
clear
B)
Isoenthalpic done
clear
C)
Isothermal done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer32)
In which of the following \[\Delta E=\Delta H\] [MP PMT 2001]
A)
\[{{N}_{2}}{{O}_{4}}(g)\] ⇌ \[2N{{O}_{2}}(g)\] done
clear
B)
\[2S{{O}_{2}}(g)+{{O}_{2}}(g)\] ⇌ \[2S{{O}_{3}}(g)\] done
clear
C)
\[{{H}_{2}}(g)+{{I}_{2}}(g)\] ⇌ \[2HI(g)\] done
clear
D)
\[{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\] ⇌ \[{{H}_{2}}O(l)\] done
clear
View Solution play_arrow
-
question_answer33)
In an adiabatic expansion of an ideal gas [KCET (Med.) 2001; MH CET 2000]
A)
\[W=-\Delta E\] done
clear
B)
\[W=\Delta E\] done
clear
C)
\[\Delta E=0\] done
clear
D)
\[W=0\] done
clear
View Solution play_arrow
-
question_answer34)
For the reaction \[C{{H}_{3}}COOH(l)+2{{O}_{2}}(g)\] ⇌ \[2C{{O}_{2}}(g)+2{{H}_{2}}O(l)\] at \[{{25}^{o}}C\] and 1 atm. pressure, \[\Delta H=-874\,kJ.\] Then the change in internal energy \[(\Delta E)\] is ?. [Orissa JEE 2002]
A)
? 874 kJ done
clear
B)
? 871.53 kJ done
clear
C)
? 876.47 kJ done
clear
D)
+ 874 kJ done
clear
View Solution play_arrow
-
question_answer35)
In a closed insulated container, a liquid is stirred with a paddle to increase its temperature. In this process, which of the following is true [CBSE PMT 2002]
A)
\[\Delta E=W=Q=0\] done
clear
B)
\[\Delta E\ne 0,\,Q=W=0\] done
clear
C)
\[\Delta E=W\ne 0,\,Q=0\] done
clear
D)
\[\Delta E=Q\ne 0,\,W=0\] done
clear
View Solution play_arrow
-
question_answer36)
Hess law is based on [MH CET 2002]
A)
Law of conservation of mass done
clear
B)
Law of conservation of energy done
clear
C)
Enthalpy is a state function done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer37)
For the reaction, \[{{C}_{3}}{{H}_{8}}(g)+5{{O}_{2}}(g)\to \,\,3C{{O}_{2}}(g)+4{{H}_{2}}O(l)\] at constant temperature, \[\Delta H-\Delta E\] is [CBSE PMT 2003]
A)
? RT done
clear
B)
+ RT done
clear
C)
? 3RT done
clear
D)
+ 3RT done
clear
View Solution play_arrow
-
question_answer38)
According to Hess?s law, the heat of reaction depends upon [MP PMT 2003]
A)
Initial condition of reactants done
clear
B)
Initial and final conditions of reactants done
clear
C)
Intermediate path of the reaction done
clear
D)
End conditions of reactants done
clear
View Solution play_arrow
-
question_answer39)
The value of \[\Delta H-\Delta E\] for the following reaction at \[{{27}^{o}}C\] will be \[2N{{H}_{3}}(g)\,\to \,{{N}_{2}}(g)+3{{H}_{2}}(g)\] [Kerala (Med.) 2002]
A)
\[8.314\times 273\times (-2)\] done
clear
B)
\[8.314\times 300\times (-2)\] done
clear
C)
\[8.314\times 27\times (-2)\] done
clear
D)
\[8.314\times 300\times (2)\] done
clear
View Solution play_arrow
-
question_answer40)
For which of the following \[\Delta E=\Delta H\] [MP PET 2003]
A)
\[{{N}_{2}}{{O}_{4}}(g)\] ⇌ \[2N{{O}_{2}}(g)\] done
clear
B)
\[2S{{O}_{2}}(g)+{{O}_{2}}(g)\] ⇌ \[2S{{O}_{3}}(g)\] done
clear
C)
\[{{H}_{2}}(g)+C{{l}_{2}}(g)\] ⇌ \[2HCl(g)\] done
clear
D)
\[{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\] ⇌ \[{{H}_{2}}O(l)\] done
clear
View Solution play_arrow
-
question_answer41)
One mole of an ideal gas is allowed to expand freely and adiabatically into vacuum until its volume has doubled. A statement which is not true concerning this expression is [Pb. PMT 1998]
A)
\[\Delta H=0\] done
clear
B)
\[\Delta S=0\] done
clear
C)
\[\Delta E=0\] done
clear
D)
\[W=0\] done
clear
View Solution play_arrow
-
question_answer42)
At \[{{27}^{o}}C,\] one mole of an ideal gas is compressed isothermally and reversibly from a pressure of 2 atm to 10 atm. The values of \[\Delta E\] and q are \[(R=2)\] [BHU 2001]
A)
0, ? 965.84 cal done
clear
B)
? 965.84 cal, + 965.84 cal done
clear
C)
+ 865.58 cal, ? 865.58 cal done
clear
D)
? 865.58 cal, ? 865.58 cal done
clear
View Solution play_arrow
-
question_answer43)
The work done by a system is 8 joule, when 40 joule heat is supplied to it. What is the increase in internal energy of system [BHU 2001]
A)
25 J done
clear
B)
30 J done
clear
C)
32 J done
clear
D)
28 J done
clear
View Solution play_arrow
-
question_answer44)
A system absorb 600J of heat and work equivalent to 300J on its surroundings. The change in internal energy is [Pb. PMT 2004]
A)
300 J done
clear
B)
400 J done
clear
C)
500 J done
clear
D)
600 J done
clear
View Solution play_arrow
-
question_answer45)
Work done during isothermal expansion of one mole of an ideal gas from 10 atom. to 1 atm at 300K is [BHU 2004]
A)
4938.8 J done
clear
B)
4138.8 J done
clear
C)
5744.1 J done
clear
D)
6257.2 J done
clear
View Solution play_arrow
-
question_answer46)
If gas, at constant temperature and pressure expands then its [MH CET 2003]
A)
Entropy increases and then decreases done
clear
B)
Internal energy increases done
clear
C)
Internal energy remains the same done
clear
D)
Internal energy decreases done
clear
View Solution play_arrow
-
question_answer47)
For the reaction \[PC{{l}_{5}}(g)\to PC{{l}_{3}}(g)+C{{l}_{2}}(g)\] [MH CET 2004]
A)
\[\Delta H=\Delta E\] done
clear
B)
\[\Delta H>\Delta E\] done
clear
C)
\[\Delta H<\Delta E\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer48)
Enthalpy \[(H)\] is equal to [MH CET 2004]
A)
Internal energy E done
clear
B)
Product of pressure (P) and volume (V) of gas done
clear
C)
Internal energy E+PV done
clear
D)
Work (W) done by a system done
clear
View Solution play_arrow
-
question_answer49)
\[\Delta {{E}^{o}}\] of combustion of isobutylene is \[-X\,kJ\,mo{{l}^{-1}}\]. The value of \[\Delta {{H}^{o}}\] is [DCE 2004]
A)
\[=\Delta {{E}^{o}}\] done
clear
B)
\[>\Delta {{E}^{o}}\] done
clear
C)
\[=0\] done
clear
D)
\[<\Delta {{E}^{o}}\] done
clear
View Solution play_arrow
-
question_answer50)
One mole of an ideal gas is allowed to expand reversibly and adibatically from a temperature of \[{{27}^{o}}C\]. If the work done during the process is \[3\,kJ\], then final temperature of the gas is \[({{C}_{V}}=20\,J/K)\] [Pb. CET 2002]
A)
100 K done
clear
B)
150 K done
clear
C)
195 K done
clear
D)
255 K done
clear
View Solution play_arrow
-
question_answer51)
Which of the following is correct regarding the internal energy of a substance [Pb. CET 2002]
A)
It is a state function done
clear
B)
It increases with increase in temperature done
clear
C)
It can be calculated by experiment done
clear
D)
It remains unaffected with change in temperature done
clear
View Solution play_arrow
-
question_answer52)
For the gaseous reaction, \[{{N}_{2}}{{O}_{4}}\to 2N{{O}_{2}}\] [Pb. CET 2003]
A)
\[\Delta H<\Delta E\] done
clear
B)
\[\Delta H=\Delta E\] done
clear
C)
\[\Delta H=0\] done
clear
D)
\[\Delta H>\Delta E\] done
clear
View Solution play_arrow
-
question_answer53)
\[2C+{{O}_{2}}\to 2CO;\,\,\Delta H=-220\,kJ\] Which of the following statement is correct for this reaction [BVP 2004]
A)
Heat of combustion of carbon is 110 kJ done
clear
B)
Reaction is exothermic done
clear
C)
Reaction needs no initiation done
clear
D)
All of these are correct done
clear
View Solution play_arrow
-
question_answer54)
An ideal gas expands in volume from \[1\times {{10}^{-3}}{{m}^{3}}\] to \[1\times {{10}^{-2}}{{m}^{3}}\] at 300 K against a constant pressure of \[1\times {{10}^{5}}N{{m}^{-2}}\]. The work done is [AIEEE 2004]
A)
270 kJ done
clear
B)
?900 kJ done
clear
C)
?900 J done
clear
D)
900 kJ done
clear
View Solution play_arrow
-
question_answer55)
Internal energy is [AFMC 2004]
A)
Partly potential and partly kinetic done
clear
B)
Totally kinetic done
clear
C)
Totally potential done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer56)
For which of the reaction \[\Delta H=\Delta E\] [MP PET 2004]
A)
\[{{N}_{2}}+3{{H}_{2}}\]⇌\[2N{{H}_{3}}\] done
clear
B)
\[{{N}_{2}}+{{O}_{2}}\to 2NO\] done
clear
C)
\[2Na+C{{l}_{2}}\to 2NaCl\] done
clear
D)
\[PC{{l}_{5}}\to PC{{l}_{3}}+C{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer57)
The work done during the expansion of a gas from a volume of \[4d{{m}^{3}}\] to \[6d{{m}^{3}}\] against a constant external pressure of \[3atm\] is (\[1L\,atm\,=101.32\,J\]) [CBSE PMT 2004]
A)
+ 304 J done
clear
B)
?304 J done
clear
C)
? 6 J done
clear
D)
?608 J done
clear
View Solution play_arrow
-
question_answer58)
For the reaction, \[{{A}_{(s)}}+2{{B}_{(g)}}\to 4{{C}_{(s)}}+{{D}_{(l)}}\]. \[\Delta H\] and \[\Delta U\] are related as ??? [Orissa JEE 2004]
A)
\[\Delta H=\Delta U\] done
clear
B)
\[\Delta H=\Delta U+3RT\] done
clear
C)
\[\Delta H=\Delta U+RT\] done
clear
D)
\[\Delta H=\Delta U-3RT\] done
clear
View Solution play_arrow
