A) 8
B) 4
C) 9
D) 3
Correct Answer: B
Solution :
[a] Initial At eqm.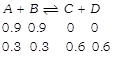 \[{{K}_{c}}=\frac{0.6\times 0.6}{0.3\times 0.3}=4\]
\[{{K}_{c}}=\frac{0.6\times 0.6}{0.3\times 0.3}=4\]
You need to login to perform this action.
You will be redirected in
3 sec
