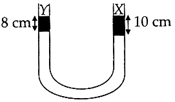
| A liquid X of density \[3.36\,\text{g}\,\text{c}{{\text{m}}^{-3}}\]is poured in a U-tube, which contains Hg. |
| Another liquid Y is poured in left arm with height 8 cm, upper levels of X and Y are same. |
| What is the density of V? |
A) \[0.8\,\text{g}\,\text{c}{{\text{m}}^{-3}}\]
B) \[1.2\,\text{g}\,\text{c}{{\text{m}}^{-3}}\]
C) \[1.4\,\text{g}\,\text{c}{{\text{m}}^{-3}}\]
D) \[1.6\,\text{g}\,\text{c}{{\text{m}}^{-3}}\]
Correct Answer: A
Solution :
[a]: As shown in adjacent figure, in the two arms of a tube pressure remains same on surface PP'.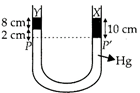 Hence, \[8\times {{\rho }_{Y}}\times g+2\times {{\rho }_{Hg}}\times g=10\times {{\rho }_{X}}\times g\] \[\therefore \]\[8{{\rho }_{Y}}+2\times 13.6=10\times 3.36\] or\[{{\rho }_{Y}}=\frac{33.6-27.2}{8}=0.8g\,c{{m}^{-3}}\]
Hence, \[8\times {{\rho }_{Y}}\times g+2\times {{\rho }_{Hg}}\times g=10\times {{\rho }_{X}}\times g\] \[\therefore \]\[8{{\rho }_{Y}}+2\times 13.6=10\times 3.36\] or\[{{\rho }_{Y}}=\frac{33.6-27.2}{8}=0.8g\,c{{m}^{-3}}\]
You need to login to perform this action.
You will be redirected in
3 sec
