A) Tetrahedral
B) square pyramidal
C) T-shape
D) square planar
Correct Answer: C
Solution :
In \[XeO{{F}_{2}}\] there are 3 bond pairs and 2 lone pair of electrons. So, the hybridisation is \[s{{p}^{3}}d\] and structure should be trigonal bipyramidal. But its actual geometry is T-shaped because of the presence of two lone pairs which occupy equatorial position.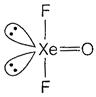
You need to login to perform this action.
You will be redirected in
3 sec
