A) \[4,2,0,+\,1/2\]
B) \[4,\text{ }2,\text{+1},+1/2\]
C) \[{{N}_{2}}\]
D) All are correct
Correct Answer: B
Solution :
| \[p=3{{s}^{2}}3{{p}^{3}}\] |
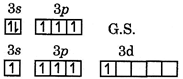 |
| In excited state spin quantum no. of last \[{{e}^{-}}\] will be\[(-1/2),\] To obey Pauli's exclusion principle |
You need to login to perform this action.
You will be redirected in
3 sec
