A) \[C{{H}_{3\,}}CHO\,\,and\,\,C{{H}_{3}}\,Br\]
B) \[Br\,C{{H}_{2}}CHO\,\,and\,\,C{{H}_{3}}OH\]
C) \[Br\,C{{H}_{2}}-C{{H}_{2}}-OC{{H}_{3}}\]
D) \[C{{H}_{3}}-CHBr-OC{{H}_{3}}\]
Correct Answer: D
Solution :
[d] \[C{{H}_{3}}-CHBr-OC{{H}_{3}}\] Addition of a proton to the terminal \[C{{H}_{2}}\] gives a carbonation which is stabilized by +R effect of \[OC{{H}_{3}}\] group.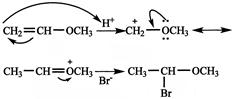
You need to login to perform this action.
You will be redirected in
3 sec
