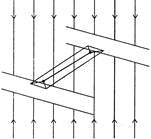
An iron block of sides \[5cm\times 8cm\times 25cm\] has to be pushed along the floor. The force required will be minimum when the surface in contact with ground is:
A)
force is the same for all surfaces
done
clear
B)
\[8\,cm\times 5\,cm\] surface
done
clear
C)
\[5\,cm\times 15\,cm\] surface
done
clear
D)
\[8\,cm\times 15\,cm\] surface
done
clear
View Answer play_arrow
A bullet is fired and get embedded in a block kept on a table. If table is frictionless, then:
A)
Potential energy gets conserved.
done
clear
B)
Kinetic energy gets conserved
done
clear
C)
Momentum gets conserved.
done
clear
D)
both (B) and (C)
done
clear
View Answer play_arrow
A bomber plane is moving horizontally with a speed of \[500m/s\] and bomb released from it, strikes the ground in 10 seconds. Angle at which it strikes the ground is \[(g=10m/{{s}^{2}})\]:
A)
\[{{\sin }^{-1}}\frac{1}{5}\]
done
clear
B)
\[ta{{n}^{-1}}\frac{1}{5}\]
done
clear
C)
\[ta{{n}^{-1}}\,5\]
done
clear
D)
\[ta{{n}^{-1}}\,1\]
done
clear
View Answer play_arrow
A force \[\vec{F}=3\hat{i}+c\hat{j}+2\hat{k}\] acting on a particle cause a displacement \[\vec{S}=-4\hat{i}+2\hat{j}+3\hat{k}\] in its own direction. If the work done is 6J, then the value of c will be:
A)
12
done
clear
B)
6
done
clear
C)
1
done
clear
D)
0
done
clear
View Answer play_arrow
The average force necessary to stop a bullet of mass \[20g\] and \[250\text{ }m/s\] as it penetrates wood to a distance of \[12cm,\] is:
A)
\[5.2\times {{10}^{3}}N\]
done
clear
B)
\[4.2\times {{10}^{3}}N\]
done
clear
C)
\[3.2\times {{10}^{3}}N\]
done
clear
D)
\[2.2\times {{10}^{3}}N\]
done
clear
View Answer play_arrow
Waterfalls from a height 500m. What is raise in temprature of water at the bottom. If the whole energy remain water. (Specific heat of water \[4200\,J/k{{g}^{o}}C\]).
A)
\[{{0.19}^{o}}C\]
done
clear
B)
\[{{0.24}^{o}}C\]
done
clear
C)
\[{{0.40}^{o}}C\]
done
clear
D)
\[{{1.16}^{o}}C\]
done
clear
View Answer play_arrow
\[7\mu f\]capacitor is charged by a 50V battery. Plow much electrostatic energy is stored by it?
A)
\[1.7\times {{10}^{-8}}J\]
done
clear
B)
\[13.6\times {{10}^{-9}}J\]
done
clear
C)
\[8.75\times {{10}^{-3}}J\]
done
clear
D)
\[6.7\times {{10}^{-7}}J\]
done
clear
View Answer play_arrow
Parallel plate condenser with oil between the plates (dielectric constant of oil \[K=2\]) has a capacitance C. if the oil is removed then capacitance of the capacitor becomes:
A)
\[\frac{C}{2}\]
done
clear
B)
\[\frac{C}{\sqrt{2}}\]
done
clear
C)
\[2C\]
done
clear
D)
\[\sqrt{2}C\]
done
clear
View Answer play_arrow
If a wire of resistance R is stretched to double its length, then the new resistance is:
A)
\[4R\]
done
clear
B)
\[2R\]
done
clear
C)
R
done
clear
D)
none of these
done
clear
View Answer play_arrow
A wave of frequency \[100Hz\] travels along a string towards its fixed ends. When this wave travels back after reflection, a node is formed at a distance of 10cm from the fixed end. The speed of the wave (incident and reflected) is:
A)
\[40\,m/s\]
done
clear
B)
\[20\,m/s\]
done
clear
C)
\[10\,m/s\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
A positively charged particles due east enters a region of uniform magnetic field directed vertically upward. This particle will:
A)
move in a circular path with a uniform speed.
done
clear
B)
move in a circular path with a decreased speed.
done
clear
C)
move in a circular path with a increased speed.
done
clear
D)
get deflected in a verticle upward direction.
done
clear
View Answer play_arrow
The peak value of A.C. voltage on a 220 volt mains will be:
A)
\[240\,\sqrt{2}\] volt
done
clear
B)
\[230\,\sqrt{2}\] volt
done
clear
C)
\[220\,\sqrt{2}\] volt
done
clear
D)
zero
done
clear
View Answer play_arrow
The ratio of charge to potential of a body is called as:
A)
Resistance
done
clear
B)
Inductance
done
clear
C)
Conductance
done
clear
D)
Capacitance
done
clear
View Answer play_arrow
In an electromagnetic wave, the electric and magnetising fields are \[100\text{ }V/m\] and\[0.265\text{ }A/m\]. The maximum energy flow will be:
A)
\[76.5\,W/{{m}^{2}}\]
done
clear
B)
\[46.7\,W/{{m}^{2}}\]
done
clear
C)
\[36.5\,W/{{m}^{2}}\]
done
clear
D)
\[26.5\,W/{{m}^{2}}\]
done
clear
View Answer play_arrow
Two identical galvanometers arc converted into ammeter and milliammeter shunts, which has more resistance due to current passing through the milliammeter is:
A)
zero
done
clear
B)
less
done
clear
C)
more
done
clear
D)
equal
done
clear
View Answer play_arrow
Radius of the circular path of and electron subjected to a perpendicular magnetic field is:
A)
\[\frac{mV}{Be}\]
done
clear
B)
\[\frac{me}{B}\]
done
clear
C)
\[\frac{mE}{B}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
Electron beam is allowed to pass normally through mutually perpendicular magnetic electric fields. If magnetic field induction and electric field strength are \[0.0004\text{ }Wb/{{m}^{2}}\] and \[3000\text{ }V/m\] respectively and beam suffers no deflection. Then the velocity of electron will be:
A)
\[1.2\times {{10}^{6}}\text{ }m/s\]
done
clear
B)
\[7.5\times {{10}^{12}}\text{ }m/s\]
done
clear
C)
\[7.5\times {{10}^{6}}\text{ }m/s\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
If the vertical components of earth's magnetic field be \[6\times {{10}^{-5}}Wb/{{m}^{2}}\] then what will be the induced potential difference produced between the rails of metre gauge when a train is running on them with speed of\[36\text{ }km/hr\]?
A)
\[9\times {{10}^{4}}\]volt
done
clear
B)
\[6\times {{10}^{-4}}\] volt
done
clear
C)
\[3\times {{10}^{-4}}\] volt
done
clear
D)
\[2\times {{10}^{4}}\] volt
done
clear
View Answer play_arrow
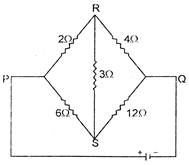
The equivalent resistance Rpq between the points P and Q is:
A)
\[8\,\Omega \]
done
clear
B)
\[1.5\,\Omega \]
done
clear
C)
\[4.5\,\Omega \]
done
clear
D)
\[6\,\Omega \]
done
clear
View Answer play_arrow
Dimensions of bubble constant are:
A)
\[[ML{{T}^{-1}}]\]
done
clear
B)
\[[ML{{T}^{4}}]\]
done
clear
C)
\[[{{M}^{o}}{{L}^{o}}{{T}^{-2}}]\]
done
clear
D)
\[[{{T}^{-1}}]\]
done
clear
View Answer play_arrow
Two racing car of masses \[{{m}_{1}}\] and \[{{m}_{2}}\] are moving in a circle of radius \[{{r}_{1}}\] and \[{{r}_{2}}\]respectively their speeds are such that each makes a complete circle in the same time t the ratio of the angular speed of the first to the second car will be:
A)
\[{{m}_{1}}{{m}_{2}}:{{r}_{1}}{{r}_{2}}\]
done
clear
B)
\[1:1\]
done
clear
C)
\[{{m}_{1}}:{{m}_{2}}\]
done
clear
D)
\[{{r}_{1}}:{{r}_{2}}\]
done
clear
View Answer play_arrow
The kinetic energy of one gramme molecule of a gas, at normal temperature and pressure will be \[(R=8.31\,J\,mole\,K)\]:
A)
\[3.4\times {{10}^{3}}J\]
done
clear
B)
\[2.7\times {{10}^{2}}J\]
done
clear
C)
\[1.2\times {{10}^{2}}J\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
Two rods having thermal conductivity in the ratio of \[5:3\] having same length area equal cross-sectional area are join by face to face. If the temperature of the free end of the first rod is \[{{100}^{o}}C\] and the temperature of free end of the second rod is \[{{20}^{o}}C\]. Then temperature of junction will be:
A)
\[{{85}^{o}}C\]
done
clear
B)
\[{{90}^{o}}C\]
done
clear
C)
\[{{50}^{o}}C\]
done
clear
D)
\[{{70}^{o}}C\]
done
clear
View Answer play_arrow
The wavelength of a light emitting from second orbit to the first orbit in a hydrogen atom will be:
A)
\[1.215\times {{10}^{-7}}m\]
done
clear
B)
\[1.215\times {{10}^{-3}}m\]
done
clear
C)
\[1.215\times {{10}^{-5}}m\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
If length of a closed organ pipe is \m and velocity of sound is\[330\text{ }mis\]. Then, the frequency of second note will be:
A)
\[2\times \frac{4}{330}\,Hz\]
done
clear
B)
\[2\times \frac{330}{4}\,Hz\]
done
clear
C)
\[3\times \frac{330}{4}\,Hz\]
done
clear
D)
\[4\times \frac{330}{4}\,Hz\]
done
clear
View Answer play_arrow
The coefficient of volumetric expansion of mercury is\[1.8\times {{10}^{-5}}^{o}C\]. The thermometer bulb has a velocity of \[{{10}^{-6}}{{m}^{3}}\] and cross-section of stern\[0.002\,c{{m}^{2}}\]. Assuming that bulb is filled with mercury at \[{{0}^{o}}C,\] the length of mercury column at \[{{100}^{o}}C\] will be:
A)
\[18\,mm\]
done
clear
B)
\[9\,mm\]
done
clear
C)
\[18\,cm\]
done
clear
D)
\[9\,cm\]
done
clear
View Answer play_arrow
A stone is tied to one end of a string of \[40\,cm\] long is whirled in a horizontal circle with a constant speed. If the stone makes 10 revolutions in 20 second. The magnitude of the acceleration is:
A)
\[990cm/{{s}^{2}}\]
done
clear
B)
\[394\,cm/{{s}^{2}}\]
done
clear
C)
\[250\,cm/{{s}^{2}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
Rectification is the process of:
A)
Low DC into high DC
done
clear
B)
Low AC into high AC
done
clear
C)
AC into DC
done
clear
D)
DC into AC
done
clear
View Answer play_arrow
A permanent magnet attracts:
A)
only paramagnetic substance
done
clear
B)
all substance
done
clear
C)
only ferromagnetic substance
done
clear
D)
all of these
done
clear
View Answer play_arrow
The resistance of a galvanometer is 60. and it can measure a maximum current of 2amp, then required shunt resistance to convert it into an ammeter reads upto 6 amp, will be:
A)
50
done
clear
B)
40
done
clear
C)
30
done
clear
D)
20
done
clear
View Answer play_arrow
As the intensity of incident light increases:
A)
Kinetic energy of emitted photoelectron
done
clear
B)
Kinetic energy of emitted photoelectron increases
done
clear
C)
Photoelectric current decreases
done
clear
D)
Photoelectric current increases
done
clear
View Answer play_arrow
A semiconductors is damaged by a strong current, due to:
A)
lack of free electrons
done
clear
B)
decrease in electrons
done
clear
C)
excess of electrons
done
clear
D)
cither (A) and (B)
done
clear
View Answer play_arrow
A proton and a-particle are accelerated through the same potential difference. The ratio of wavelength of the proton to that of an a-particle is:
A)
\[2\sqrt{2}:1\]
done
clear
B)
\[1:2\sqrt{2}\]
done
clear
C)
\[2:1\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
A chain reaction in a fission of uranium is possible because:
A)
fragments in the fission are radioactive
done
clear
B)
more than one neutron is given out
done
clear
C)
small amount of energy is released
done
clear
D)
large amount of energy is released
done
clear
View Answer play_arrow
The x-ray cannot be diffracted by means of an extraordinary grating because of:
A)
large wavelength
done
clear
B)
high speed
done
clear
C)
short wavelength
done
clear
D)
low speed
done
clear
View Answer play_arrow
In a coil having emf of 8 volt is induced for \[0.05\] the value of current changes from 2amp to 4amp. What will be the value of coefficient of self induction of the coil?
A)
\[0.8\] henry
done
clear
B)
\[0.4\] henry
done
clear
C)
\[0.2\] henry
done
clear
D)
none of these
done
clear
View Answer play_arrow
The amplification factor of a triode valve depends upon:
A)
relative position of cathode grid and plate
done
clear
B)
plate potential and grid potential
done
clear
C)
temperature of the anode
done
clear
D)
ternperature of the cathode
done
clear
View Answer play_arrow
Dark lines in the solar spectrum are due to:
A)
absorption of certain wavelengths, producing dark lines in the solar system
done
clear
B)
absence of certain elements in the sun.
done
clear
C)
black body radiations from the sun
done
clear
D)
scattering of light
done
clear
View Answer play_arrow
The astronomical microscope consists of objective and eyepiece. The focal length of the objective is:
A)
five times shorter than of eyepiece
done
clear
B)
shorter than that of eyepiece
done
clear
C)
greater than that of eyepiece
done
clear
D)
equal to that of eyepiece
done
clear
View Answer play_arrow
Objective with large aperture are used in telescope for:
A)
reducing less aberration
done
clear
B)
ease of manufacture
done
clear
C)
greater resolution
done
clear
D)
greater magnification
done
clear
View Answer play_arrow
A jet plane emits sound with a frequency 990 cycles/sound and carries on board a source of light emitting radiation of wavelength\[4500\overset{o}{\mathop{A}}\,\]. If the jet plane approaches the airport with the velocity 1080 &/n/hour, then the frequency of sound beared by the observer on airport will be:
A)
\[11890\text{ }cycles/sec.\]
done
clear
B)
\[10890\text{ }cycles/sec.\]
done
clear
C)
\[12890\text{ }cycles/sec.\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
When a slow neutron captured by a \[{{U}^{235}}\]nucleus a fission results which release 200 MeV energy. Then the number of fission per second of nclei undergoing fission is:
A)
\[5\times {{10}^{10}}\]
done
clear
B)
\[5\times {{10}^{12}}\]
done
clear
C)
\[5\times {{10}^{4}}\]
done
clear
D)
\[5\times {{10}^{10}}\]
done
clear
View Answer play_arrow
If one mole of monoatomic gas \[\gamma =\frac{5}{3}\] is mixed with one mole of diatomic gas \[\gamma =\frac{7}{5}\]. The value of \[\gamma \] for the mixtures is:
A)
\[2.5\]
done
clear
B)
\[1.5\]
done
clear
C)
\[1.8\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
What is the ratio of specific charge elm of an a-particles and that of electron (Proton is 1840 times heavier than an electron)?
A)
\[\frac{1}{3680}\]
done
clear
B)
\[\frac{1}{4680}\]
done
clear
C)
\[\frac{1}{5680}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
A prism of \[{{6}^{o}}\] is made of material of refractive index \[\frac{5}{3}\]. The deviation caused by it is:
A)
\[{{2}^{o}}\]
done
clear
B)
\[{{8}^{o}}\]
done
clear
C)
\[{{4}^{o}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
Two flasks X and Y have capacity 1L and 2L respectively and each of them contains 1 mole of a gas. The temperatures of the flasks are so adjusted that average speed of molecules in X is twice as those in Y. The pressure in flasks X would be
A)
same as that in Y
done
clear
B)
half of that in Y
done
clear
C)
twice of that in Y
done
clear
D)
Eight times of that in Y
done
clear
View Answer play_arrow
Which one of the following set of quantum numbers is not possible for 4p electron?
A)
\[n=4,\,l=1,\,m=-1,\,{{m}_{s}}=+\frac{1}{2}\]
done
clear
B)
\[n=4,\,l=1,\,m=0,\,{{m}_{s}}=+\frac{1}{2}\]
done
clear
C)
\[n=4,\,l=1,\,m=2,\,{{m}_{s}}=+\frac{1}{2}\]
done
clear
D)
\[n=4,\,l=1,\,m=-1,\,{{m}_{s}}=-\frac{1}{2}\]
done
clear
View Answer play_arrow
How many unit cells are present in a cube shaped ideal crystal of \[NaCl\] of mass\[1.00\text{ }g\]? [Atomic masses : \[Na=23,Cl=35.51\]]
A)
\[5.14\times {{10}^{21}}\]
done
clear
B)
\[1.28\times {{10}^{21}}\]
done
clear
C)
\[1.71\times {{10}^{21}}\]
done
clear
D)
\[2.57\times {{10}^{21}}\]
done
clear
View Answer play_arrow
Which of the following molecule exhibit maximum enol content?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following compounds give oxygen on heating?
A)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}},\,\,KCl{{O}_{3}}\]
done
clear
B)
\[{{(N{{H}_{4}})}_{2}}C{{r}_{2}}{{O}_{7}},KCl{{O}_{3}}\]
done
clear
C)
\[Pb{{(N{{O}_{3}})}_{2}},\,AgN{{O}_{3}}\]
done
clear
D)
Both (A) and (C)
done
clear
View Answer play_arrow
Beckmann transformation of
followed by hydrolysis will yield
A)
ben zoic acid and benzylaminc
done
clear
B)
phony [acetic acid and benzylamine
done
clear
C)
aniline and phenyl acetic acid
done
clear
D)
benxoic acid and aniline
done
clear
View Answer play_arrow
The correct increasing bond angles order is:
A)
\[B{{F}_{3}}<N{{F}_{3}}<P{{F}_{3}}<Cl{{F}_{3}}\]
done
clear
B)
\[Cl{{F}_{3}}<P{{F}_{3}}<N{{F}_{3}}<B{{F}_{3}}\]
done
clear
C)
\[B{{F}_{3}}\approx N{{F}_{3}}<P{{F}_{3}}<Cl{{F}_{3}}\]
done
clear
D)
\[B{{F}_{3}}<N{{F}_{3}}<P{{F}_{3}}<Cl{{F}_{3}}\]
done
clear
View Answer play_arrow
Product
A)
done
clear
B)
done
clear
C)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
The increasing thermal stability of the hydrides of group 16 follows the sequence
A)
\[{{H}_{2}}O,\,{{H}_{2}}S,\,{{H}_{2}}Se,\,{{H}_{2}}Te\]
done
clear
B)
\[{{H}_{2}}Te,{{H}_{2}}Se,\,{{H}_{2}}S,\,{{H}_{2}}O\]
done
clear
C)
\[{{H}_{2}}S,{{H}_{2}}O,{{H}_{2}}Se,\,\,{{H}_{2}}Te\]
done
clear
D)
\[{{H}_{2}}Se,\,{{H}_{2}}S,{{H}_{2}}O,\,{{H}_{2}}Te\]
done
clear
View Answer play_arrow
Dimethyl glyoxime in a suitable solvent was refluxed for 10 min with pure pieces of nickel sheet, it will result in
A)
red preciptate
done
clear
B)
blue precipitate
done
clear
C)
yellow precipitate
done
clear
D)
no precipitate
done
clear
View Answer play_arrow
The hybrid state of \[Co\] in high spin \[{{K}_{3}}Co{{F}_{6}}\] complex is
A)
\[s{{p}^{3}}{{d}^{2}}\]
done
clear
B)
\[s{{p}^{3}}\]
done
clear
C)
\[{{d}^{2}}s{{p}^{3}}\]
done
clear
D)
\[s{{p}^{3}}d\]
done
clear
View Answer play_arrow
End product of the following reaction is._____ by mechanism,_____
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following is a biodegradable polymer?
A)
Polythene
done
clear
B)
Bakelite
done
clear
C)
PHBV
done
clear
D)
PVC
done
clear
View Answer play_arrow
Which of the following statement is not true?
A)
Some disinfectants can be used as antiseptic at low concentration.
done
clear
B)
Noretbindrone is a pheromone.
done
clear
C)
Aspirin is analgesic and antipyretic
done
clear
D)
[Su] phadiazine is a synthetic antibacterial.
done
clear
View Answer play_arrow
Which one of the following compounds is used as a body deodorant?
A)
Aspirin
done
clear
B)
Omeprazole
done
clear
C)
Bithional
done
clear
D)
P- chlorometaxylenol
done
clear
View Answer play_arrow
Among the following the molecule with highest dipole moment is
A)
\[C{{H}_{3}}Cl\]
done
clear
B)
\[C{{H}_{2}}C{{l}_{2}}\]
done
clear
C)
\[CHC{{l}_{3}}\]
done
clear
D)
\[CC{{l}_{4}}\]
done
clear
View Answer play_arrow
The cyanide ion, \[C{{N}^{-}}\] and \[{{N}_{2}}\] are isoelectronic. But in contrast to \[C{{N}^{-}},{{N}_{2}}\] is chemically inert, because of
A)
low bond energy
done
clear
B)
absence of bond polarity
done
clear
C)
unsymmetrical electron distribution
done
clear
D)
presence of more number of electrons in bonding orbitals
done
clear
View Answer play_arrow
Solute A is a ternary electrolyte and solute B is a non-eletrolyte. If \[0.1\text{ }M\] solution of solute B produces an osmotic pressure of 2P, then \[0.05\text{ }M\]solution of A at the same temperature will produce an osmotic pressure equal to
A)
\[1.5\,P\]
done
clear
B)
\[2\,P\]
done
clear
C)
\[3\,P\]
done
clear
D)
P
done
clear
View Answer play_arrow
Which of the following relationships is correct?
A)
\[K=\frac{T\Delta {{S}^{o}}-\Delta {{H}^{o}}}{RT}\]
done
clear
B)
\[K=\frac{\Delta {{H}^{o}}-T\Delta {{S}^{o}}}{RT}\]
done
clear
C)
In \[K=\frac{T\Delta {{S}^{o}}-\Delta {{H}^{o}}}{RT}\]
done
clear
D)
In \[K=\frac{\Delta {{H}^{o}}-T\Delta {{S}^{o}}}{RT}\]
done
clear
View Answer play_arrow
If \[{{K}_{p}}\] for a reaction \[{{A}_{(g)}}+2{{B}_{(g)}}3{{C}_{(g)}}+{{D}_{(g)}}\] is \[0.05\] atm at \[1000K.\] Its \[{{K}_{C}},\] in terms of R will be
A)
\[20000\text{ }R\]
done
clear
B)
\[0.02\text{ }R\]
done
clear
C)
\[5\times {{10}^{-5}}\,R\]
done
clear
D)
\[\frac{5\times {{10}^{-5}}}{R}\]
done
clear
View Answer play_arrow
When \[NaCl\] is added to the reaction mixture of an oil and caustic soda, the soap is thrown out because
A)
\[NaCl\] is an ionic compound
done
clear
B)
soap is insoluble in the presence of chloride ions
done
clear
C)
the solubility product of \[NaCl\] decreases in the presence of soap.
done
clear
D)
the solubility of the soap is exceeded due to the increased concentration of \[N{{a}^{+}}\] ions.
done
clear
View Answer play_arrow
Thermal decomposition method is used to purify
A)
\[Ni\]
done
clear
B)
\[Cr\]
done
clear
C)
\[Sn\]
done
clear
D)
\[Pb\]
done
clear
View Answer play_arrow
Which of the following does not contain P-O-P bond?
A)
Isohypophosphoric acid
done
clear
B)
Diphosphorus acid
done
clear
C)
Diphosphoric acid
done
clear
D)
Hypophosphoric acid
done
clear
View Answer play_arrow
A compound formed by elements A and B has a cubic structure in which A atoms are at the corners of the cube and B atoms are at the face centres. The formula for the compound is
A)
\[{{A}_{2}}{{B}_{3}}\]
done
clear
B)
\[A{{B}_{3}}\]
done
clear
C)
\[{{A}_{3}}B\]
done
clear
D)
\[{{A}_{3}}{{B}_{2}}\]
done
clear
View Answer play_arrow
The vapour pressure of pure liquid solvent A is \[0.80\] atm. When a non-volatile substance B is added to the solvent, its vapour pressure drops to \[0.60\]atm. What is the mole fraction of component B is the solution?
A)
\[0.75\]
done
clear
B)
\[0.25\]
done
clear
C)
\[0.40\]
done
clear
D)
\[0.60\]
done
clear
View Answer play_arrow
Calculate the standard free energy change for the reaction. \[2Ag+2{{H}^{+}}\xrightarrow{{}}{{H}_{2}}+2A{{g}^{+}}\] \[{{E}^{o}}\] for \[A{{g}^{+}}+c\xrightarrow{{}}Ag\]is \[0.80V\]
A)
\[+154.4\text{ }KJ\]
done
clear
B)
\[+308.8\text{ }KJ\]
done
clear
C)
\[-154.4\text{ }KJ\]
done
clear
D)
\[-308.8\text{ }KJ\]
done
clear
View Answer play_arrow
In the cell \[Zn|Z{{n}^{2+}}({{C}_{1}})||C{{u}^{2+}}({{C}_{2}})|Cu\,\,{{E}_{cell}}-{{E}^{o}}_{cell}=0.0591\,V\] The ratio \[{{C}_{1}}/{{C}_{2}}\] at 298K will be
A)
\[2\]
done
clear
B)
\[100\]
done
clear
C)
\[{{10}^{-2}}\]
done
clear
D)
\[1\]
done
clear
View Answer play_arrow
Type of isomerism shown by \[[Cr{{(N{{H}_{3}})}_{5}}N{{O}_{2}}]C{{l}_{2}}\] is
A)
optical
done
clear
B)
iomsation
done
clear
C)
geometrical
done
clear
D)
linkage
done
clear
View Answer play_arrow
\[C{{e}^{3+}},L{{a}^{3+}},P{{m}^{3+}}\] and \[Y{{b}^{3+}}\] have ionic radii in the increasing order as
A)
\[L{{a}^{3+}}<C{{e}^{3+}}<P{{m}^{3+}}<Y{{b}^{3+}}\]
done
clear
B)
\[Y{{b}^{3+}}<P{{m}^{3+}}<C{{e}^{3+}}<L{{a}^{3+}}\]
done
clear
C)
\[L{{a}^{3+}}=C{{e}^{3+}}<P{{m}^{3+}}<Y{{b}^{3+}}\]
done
clear
D)
\[Y{{b}^{3+}}<P{{m}^{3+}}<L{{a}^{3+}}<C{{e}^{3+}}\]
done
clear
View Answer play_arrow
Alum is widely used to purify water since
A)
it forms complex with clay particles
done
clear
B)
it coagulates the mud particles
done
clear
C)
it exchanges \[C{{a}^{2+}}\] and \[M{{g}^{2+}}\] ions present in hard water.
done
clear
D)
its sulphate ion is water purifier
done
clear
View Answer play_arrow
In the given reaction, X is \[C{{H}_{3}}CHO+C{{H}_{2}}{{(COOH)}_{2}}\xrightarrow[heat]{Pyridine}X\]
A)
\[C{{H}_{3}}COOH\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}COOH\]
done
clear
C)
\[C{{H}_{3}}CH=CH.COOH\]
done
clear
D)
\[HOOC.CH=CH.COOH\]
done
clear
View Answer play_arrow
If \[S{{O}_{2}},\] content in the atmosphere is \[0.12\text{ }ppm\] by volume, pH of rain water (assume 1.00% ionisation of acid rain as monobasic acid) is
A)
\[5.7\]
done
clear
B)
\[5.6\]
done
clear
C)
\[5.4\]
done
clear
D)
\[2.0\]
done
clear
View Answer play_arrow
A deliquescent white crystalline hydroxide X reacts with a nitrate Y to form another hydroxide which decomposes to give a insoluble brown layer of its oxide. X is a powerful cautery and breaks down the proteins of skin flesh to a pasty mass. X and Y are
A)
\[NaOH,AgN{{O}_{3}}\]
done
clear
B)
\[NaOH,Zn{{(N{{O}_{3}})}_{2}}\]
done
clear
C)
\[NaOH,Al{{(N{{O}_{3}})}_{3}}\]
done
clear
D)
\[Ca{{(OH)}_{2}},\,HgN{{O}_{3}}\]
done
clear
View Answer play_arrow
Mark out the wrong statement about vander waal?s constant 'a'
A)
'a' reflects magnitude of intermolecular forces.
done
clear
B)
Lower the value of 'a' greater the attractive forces.
done
clear
C)
Higher the value of W higher the deviation from ideal gas.
done
clear
D)
Its unit is atm\[{{L}^{2}}\,mo{{l}^{-2}}.\].
done
clear
View Answer play_arrow
Carbon monoxide acts as a Lewis base because it has
A)
a double bond between C and O atoms.
done
clear
B)
a triple bond between C and O atoms.
done
clear
C)
a lone pair of electrons on the C atom
done
clear
D)
two lone pairs of electrons on the O atom.
done
clear
View Answer play_arrow
Con side the following redox reaction\[VO_{3}^{2-}+MnO_{4}^{-}\xrightarrow{{}}M{{n}^{2+}}+VO_{4}^{3-}\]1 mole of \[VO_{3}^{2-}\] is oxidised by x mole of \[MnO_{4}^{-}\]. Thus, x is
A)
\[0.2\]
done
clear
B)
\[0.4\]
done
clear
C)
\[0.8\]
done
clear
D)
\[1.0\]
done
clear
View Answer play_arrow
The decomposition of \[{{N}_{2}}O\] into \[{{N}_{2}}\] and O in the presence of gaseous argon follows second order kinetics with \[=K(5.0\times {{10}^{11}}L\,mo{{l}^{-1}}\,{{S}^{-1}}){{e}^{-29000K/T}}\]Activation energy of the reaction is
A)
\[121\,KJ\,\,mo{{l}^{-1}}\]
done
clear
B)
\[241\,KJ\,\,mo{{l}^{-1}}\]
done
clear
C)
\[201\,KJ\,\,mo{{l}^{-1}}\]
done
clear
D)
\[280\,KJ\,\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
For 2s orbital the nodal surface exists at a distance of
A)
\[{{a}_{0}}\] from the nucleus
done
clear
B)
\[1.5\,{{a}_{0}}\] from the nucleus
done
clear
C)
\[2\,{{a}_{0}}\] from the nucleus
done
clear
D)
none of these is correct
done
clear
View Answer play_arrow
Which of the following is diamagnetic?
A)
Superoxide ion
done
clear
B)
Carbon molecule
done
clear
C)
Oxygen molecule
done
clear
D)
Unipositivc ion of nitrogen molecule
done
clear
View Answer play_arrow
What is the entropy change (in \[J{{K}^{-1}}\text{ }mo{{l}^{-1}}\]) when one mole of ice is converted into water at \[{{0}^{o}}C\]? (The enthalpy change for the conversion of ice to liquid water is \[6.0\text{ }KJ\text{ }mo{{l}^{-1}}\] at \[{{0}^{o}}C\])
A)
\[20.13\]
done
clear
B)
\[2.013\]
done
clear
C)
\[2.198\]
done
clear
D)
\[21.98\]
done
clear
View Answer play_arrow
To make a solution of \[pH=12,\]the amount of \[NaOH\] dissolved in one litre of the solution should be
A)
\[0.1\,g\]
done
clear
B)
\[0.2\,g\]
done
clear
C)
\[0.4\,g\]
done
clear
D)
\[1.2\,g\]
done
clear
View Answer play_arrow
Which one of the following radioisotopes is used in the treatment of blood cancer?
A)
\[{{P}^{32}}\]
done
clear
B)
\[C{{o}^{60}}\]
done
clear
C)
\[{{I}^{131}}\]
done
clear
D)
\[N{{a}^{24}}\]
done
clear
View Answer play_arrow
Collodion is a collodial solution of
A)
Sucrose in water
done
clear
B)
Cellulose in water
done
clear
C)
Cellulose nitrate in water
done
clear
D)
Cellulose nitrate in ethanol
done
clear
View Answer play_arrow
Which of the following is called inorganic graphite?
A)
\[{{B}_{3}}{{N}_{3}}{{H}_{6}}\]
done
clear
B)
\[{{B}_{3}}{{N}_{3}}\]
done
clear
C)
\[SiC\]
done
clear
D)
\[{{P}_{4}}{{S}_{3}}\]
done
clear
View Answer play_arrow
The correct IUPAC name of the spiro compound
A)
1-Oxaspiro \[[4.3]\] octane
done
clear
B)
5-oxospiro \[[3.4]\] octane
done
clear
C)
5-Oxaspiro \[[3.4]\] octane
done
clear
D)
1-oxospiro \[[4.3]\] octane
done
clear
View Answer play_arrow
Select the defining feature of living organism from the given below options.
A)
Growth, Conciousness, metabolism.
done
clear
B)
Cellular organization, growth, reproduction
done
clear
C)
Conciousness, reproduction
done
clear
D)
Metabolism, cellular organization
done
clear
View Answer play_arrow
Members of kingdom protista are primarily
A)
Chemosynthetic
done
clear
B)
Terrestrial
done
clear
C)
Aquatic, fresh water or sea water
done
clear
D)
Photo organo-heterotrophic
done
clear
View Answer play_arrow
Agaricus, claviceps, penicillium
A)
produce exogenous meiospore
done
clear
B)
develop fruiting body in sexual life cycle
done
clear
C)
have aseptate mycelium
done
clear
D)
produce predominant dikaryophase in their life cycle.
done
clear
View Answer play_arrow
TMV and \[{{T}_{2}}\] bacteriophage
A)
are without tail fibres.
done
clear
B)
have SS RNA and DS DNA respectively
done
clear
C)
have capsid around SS DNA
done
clear
D)
more than one are correct
done
clear
View Answer play_arrow
Pyrenoids as food storage body are found in
A)
Algae, Hornworts
done
clear
B)
Moss, Liverworts
done
clear
C)
Liverworts, Algae
done
clear
D)
None of these
done
clear
View Answer play_arrow
Entero coelomate invertebrates belong to
A)
Annelida
done
clear
B)
Echinodermata
done
clear
C)
Arthopoda
done
clear
D)
Mollusca
done
clear
View Answer play_arrow
Peripatus is connecting link between
A)
Annelida and Mollusca
done
clear
B)
Mollusca and Arthropoda
done
clear
C)
Annelida and Arthropoda
done
clear
D)
Coelentrata and Annelida
done
clear
View Answer play_arrow
Causative agent of bubonic plague is transmitted by
A)
Aedes
done
clear
B)
Xenopsylla
done
clear
C)
Cimex
done
clear
D)
Cockroach
done
clear
View Answer play_arrow
An animal has following characteristic features (i) Bilateral symmetry (ii) Closed circulatory system (iii) Metameric segmentation This animal belongs to
A)
Echinodermata
done
clear
B)
Mollusca
done
clear
C)
Arthropoda
done
clear
D)
Annelida
done
clear
View Answer play_arrow
Select odd one with rrespect to typical character of Nematoda.
A)
Exclusive parasite
done
clear
B)
Pseudo coel
done
clear
C)
Non living cuticle
done
clear
D)
Syncytical epidermis
done
clear
View Answer play_arrow
Ferns, Mosses, Equisetum
A)
form gamete by mitotic division
done
clear
B)
produce parasitic gametophytes
done
clear
C)
are without Archegonia
done
clear
D)
are vascular tissue bearing terrestrial plants
done
clear
View Answer play_arrow
Modified aerial stem with branching of unlimited growth as photosynthetic structure is
A)
Cylindrical in Opuntia and Euphorbia
done
clear
B)
Fleshy and cylindrical in Euphorbia
done
clear
C)
Flattened in Opuntia and Euphorbia
done
clear
D)
Cylindrical in Opuntia
done
clear
View Answer play_arrow
The fruit in Mango and Coconut
A)
is many seeded
done
clear
B)
is developed from apocarpous ovary
done
clear
C)
is developed from poly carpellary
done
clear
D)
has hard and stony endocarp
done
clear
View Answer play_arrow
Gymnosperms are plants in which the ovules are net enclosed by ovary and remain exposed. Here
A)
Roots are generally adventitious
done
clear
B)
Eustelic condition is found
done
clear
C)
Female gametophyte is parasite over diploid endosperm
done
clear
D)
Dimorphic root, stem and leaves are always found.
done
clear
View Answer play_arrow
Select the non-reducing sugar from below given options
A)
Glucose
done
clear
B)
Sucrose
done
clear
C)
Maltose
done
clear
D)
Galactose
done
clear
View Answer play_arrow
Dog fish has ampulla of Lorenzini. These are
A)
Auditory structures
done
clear
B)
Rheoreceptors
done
clear
C)
Balancing structures
done
clear
D)
Thermo receptors
done
clear
View Answer play_arrow
The bat differs from birds in having
A)
Wings
done
clear
B)
Sweat glands
done
clear
C)
Feathers
done
clear
D)
Four chambered heart
done
clear
View Answer play_arrow
Select wrong with respect to aerial adaptation of birds
A)
Sternum with keel
done
clear
B)
Single ovary
done
clear
C)
Gizzard
done
clear
D)
Pneumatic bones
done
clear
View Answer play_arrow
Cytidine is a/an
A)
nuclcotide
done
clear
B)
nuclcoside
done
clear
C)
essential ammo acid
done
clear
D)
non-essential ammo acid
done
clear
View Answer play_arrow
The first stage of mitosis is prophase. It is marked by the
A)
Different shape of chromosome
done
clear
B)
Completion of condensation of chromosomes
done
clear
C)
Starting of condensation
done
clear
D)
Reappearance of Golgi bodies and ER
done
clear
View Answer play_arrow
Match the following
Column-I
Column-II
(i) Dalton Complex
(A) Cristae
(ii) NOR
(B) SER
(iii) Cytochrome \[{{P}_{450}}\]
(C) Cisternae
(iv) Plastochondria
(D) Sec-constriction
A)
(iv)-A (iii)-B (ii)-C (i)-D
done
clear
B)
(iv)-B (iii)-A (ii)-C (i)-D
done
clear
C)
(iv)-A (iii)-B (ii)-D (i)-C
done
clear
D)
(iv)-A (iii)-D (ii)-C (i)-B
done
clear
View Answer play_arrow
Axoneme part of eukaryotic flagellum has microtubules
A)
9
done
clear
B)
27
done
clear
C)
18
done
clear
D)
20
done
clear
View Answer play_arrow
Select the wood that has high density and darker
A)
Early wood
done
clear
B)
Late wood
done
clear
C)
Spring wood
done
clear
D)
More than one option
done
clear
View Answer play_arrow
As compared to monocot root, the dicot root has/shows
A)
Fewer xylem bundles
done
clear
B)
Primary growth only
done
clear
C)
Pith large and well developed
done
clear
D)
Pericycle is sclerenchymatous
done
clear
View Answer play_arrow
Select the odd one with respect to inhibition of flowering by light break reaction
A)
Soyabean
done
clear
B)
Barley
done
clear
C)
Coffee
done
clear
D)
Rice
done
clear
View Answer play_arrow
In case of walnut, cherry and cotton, thinning is performed by
A)
BAP
done
clear
B)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
C)
IAA
done
clear
D)
ABA
done
clear
View Answer play_arrow
Select the similarity from the given below options with respect to lactic acid and alcoholic fermentation
A)
ETC
done
clear
B)
EMP Pathway
done
clear
C)
Link reaction
done
clear
D)
TCA cycle
done
clear
View Answer play_arrow
Inside a living cell, the complete break down of glucose yields
A)
Heat energy only
done
clear
B)
Energy, most of which is given in the form of ATP
done
clear
C)
Energy most of which is given out as heat
done
clear
D)
38 ATP only
done
clear
View Answer play_arrow
In photosynthetic carbon oxidation pathway
A)
Carbon loss is seen with production of ATP
done
clear
B)
\[{{O}_{2}}\] is used with synthesis of sugar
done
clear
C)
Regeneration of PGA with utilization of ATP
done
clear
D)
No production of \[C{{O}_{2}}\] and NADPH
done
clear
View Answer play_arrow
Regeneration and reduction steps of Calvin Cycle are
A)
Dependent on energy
done
clear
B)
Present in thylackoid membrane
done
clear
C)
Most crucial steps
done
clear
D)
Dependent on NADPH
done
clear
View Answer play_arrow
Select the mineral element that is a constituent of thiamine, biotin and fcrrodoxin.
A)
Iron
done
clear
B)
Sulphur
done
clear
C)
Calcium
done
clear
D)
Magnesium
done
clear
View Answer play_arrow
Active transport is different from simple diffusion in the
A)
Absence of transport protein saturation
done
clear
B)
Presence of uphill movement
done
clear
C)
Presence of downhill movement
done
clear
D)
Absence of transport protein movement
done
clear
View Answer play_arrow
Intervertebral disc is made of _______cartilage
A)
Hyaline
done
clear
B)
Fibrous
done
clear
C)
Elastic
done
clear
D)
Calcified
done
clear
View Answer play_arrow
Select the mcrocrine gland
A)
Sebaceous gland
done
clear
B)
Salivary gland
done
clear
C)
Mammary gland
done
clear
D)
Axillary sweat gland
done
clear
View Answer play_arrow
The part of cockroach's eye which is comparable to the retina of vertebrate's eye is
A)
Pigmented sheath
done
clear
B)
Crystalline cone
done
clear
C)
Corneal facet
done
clear
D)
Rhabdome
done
clear
View Answer play_arrow
Which of the following is a parasite in the seminal vesicle of earthworm?
A)
Trypanosoma
done
clear
B)
Eimeria
done
clear
C)
Monocystis
done
clear
D)
Babesia
done
clear
View Answer play_arrow
Chloragogen cells are found in
A)
Reproductive organs of earthworm
done
clear
B)
Earthworm skin
done
clear
C)
Coelomic fluid of earthworm
done
clear
D)
Blood of earthworm
done
clear
View Answer play_arrow
Most enzymes consist of two parts these are
A)
Cofactor and apoenzyme
done
clear
B)
Enzyme and apoenzyme
done
clear
C)
Cocnzymc and enzyme
done
clear
D)
Holo enzyme and prosthetics group
done
clear
View Answer play_arrow
Lac operon as well as as trp operon both
A)
arc found in bacteria
done
clear
B)
are repressible system
done
clear
C)
show positive control
done
clear
D)
are inducible system
done
clear
View Answer play_arrow
Select the odd one with respect to organic farming
A)
K-coli
done
clear
B)
Nostoc
done
clear
C)
Oscillatoria
done
clear
D)
Anabaena
done
clear
View Answer play_arrow
Cotton plant is resistant to Jassids due to presence of
A)
Hairy leaves
done
clear
B)
Solid stem
done
clear
C)
Smooth leaves
done
clear
D)
High amount of aspartic acid in stem
done
clear
View Answer play_arrow
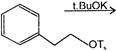
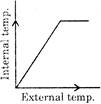
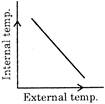
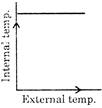
If there is an increase in external temperature, then which one of these graph is true for most of the animals and nearly all plants
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
If 16 individuals in a laboratory population of 80 fruit flies died during a specified time interval, say two weeks, the death rate in this population during this period is
A)
\[16/(80\times 2)\]
done
clear
B)
\[16\times 2/80\]
done
clear
C)
\[8\times 7/40\times 2\]
done
clear
D)
\[(80/16)\times 2\]
done
clear
View Answer play_arrow
In a particular climatic condition, decomposition rate is slower, if detritus is rich in
A)
Lignin and water soluble substances
done
clear
B)
Chitin andlignin
done
clear
C)
Nitrogen and chitin
done
clear
D)
Water soluble substance and nitrogen
done
clear
View Answer play_arrow
In terrestrial ecosystems, the pyramids of number and biomass are generally
A)
Inverted and upright respectively
done
clear
B)
Upright and. inverted respectively
done
clear
C)
Spindle shaped
done
clear
D)
Upright
done
clear
View Answer play_arrow
How many genetically different strains of rice are found in India?
A)
1000
done
clear
B)
\[<50000\]
done
clear
C)
\[>50000\]
done
clear
D)
20000
done
clear
View Answer play_arrow
The Earth summit, a historic convention on biodiversity, was held in
A)
Rio de Janeiro
done
clear
B)
Kyoto
done
clear
C)
Canada
done
clear
D)
Johannesburg
done
clear
View Answer play_arrow
Cyclosporin is
A)
used against snake venom
done
clear
B)
used as anticancer drug
done
clear
C)
used as antiaids
done
clear
D)
an immune suppressant drug
done
clear
View Answer play_arrow
Which one of these is a method of birth control?
A)
ZifT
done
clear
B)
GIFT
done
clear
C)
IUDS
done
clear
D)
IVF - ET
done
clear
View Answer play_arrow
Speed ball is a mixture of
A)
Cocainet Heroin
done
clear
B)
Ganjat Heroin
done
clear
C)
Cocainet Hashish
done
clear
D)
Cocainet Marijuana
done
clear
View Answer play_arrow
Tassar silk is extracted from
A)
Apis dorsata
done
clear
B)
Attacus ricini
done
clear
C)
Bombyx mori
done
clear
D)
Antheraea royle
done
clear
View Answer play_arrow
Select the property of acquired immunity that is the basis of vaccination
A)
Memory
done
clear
B)
Diversity
done
clear
C)
Specificity
done
clear
D)
Discrimination
done
clear
View Answer play_arrow
Parkinson disease is caused due to deficiency of
A)
Dopamine
done
clear
B)
GABA
done
clear
C)
Endorphin
done
clear
D)
Acetylcholine
done
clear
View Answer play_arrow
Passive immunity is defined as immunity
A)
Acquired through first exposure to a disease
done
clear
B)
Provided by cellular barriers against a pathogen
done
clear
C)
Achieved through the sera of other animals enriched in antibodies
done
clear
D)
Achieved through vaccination
done
clear
View Answer play_arrow
For the production of 12 polygonum types of embrayosac, how many reduction divisions are required in megasporocyte?
A)
12
done
clear
B)
3
done
clear
C)
6
done
clear
D)
11
done
clear
View Answer play_arrow
Down syndrome and KUnefclter syndrome both can be included in
A)
Allosomal trisomy
done
clear
B)
Trisomy
done
clear
C)
Monosomy
done
clear
D)
Autosomal trisomy
done
clear
View Answer play_arrow
Read the following statements carefully A. Neelakurinji flowers once in 12 years B. In majority of organisms, male and female gametes are motile C. During embryogenesis, zygote undergoes mitosis and cell differentiation
A)
All arc correct
done
clear
B)
Only (B) is incorrect
done
clear
C)
All are incorrect
done
clear
D)
(A) and (B) are correct
done
clear
View Answer play_arrow
In over 60% of angiosperms, pollen grains are shed at two celled stage. Generative cell of this stage
A)
has dense cytoplasm
done
clear
B)
floats in the nucleoplasm of the vegetative cell
done
clear
C)
is diploid
done
clear
D)
is larger than tube cell
done
clear
View Answer play_arrow
How many meiosis are required in megasporocyte to produce 12 polygonum type of embryo sacs?
A)
3
done
clear
B)
6
done
clear
C)
11
done
clear
D)
12
done
clear
View Answer play_arrow
If a cross is made between homozygous tall plant with yellow seeds (TTYY) and dwarf plant with yellow seeds (ft Yy). What proportions of tall and yellow seeded offspring could be expected in resulting generation?
A)
54%
done
clear
B)
100%
done
clear
C)
50%
done
clear
D)
6.25%.
done
clear
View Answer play_arrow
Down's syndrome and Kimefelter's syndrome both can be included in
A)
Autosomal trisomy
done
clear
B)
Monosomy
done
clear
C)
Trisomoy
done
clear
D)
Allosomal trisomy
done
clear
View Answer play_arrow
If the proportion of 5 methyl uracil in a ds-DNA is 7% then what will be the total proportion of monocyclic nitrogen bases in this DNA?
A)
50%
done
clear
B)
43%
done
clear
C)
14%
done
clear
D)
86%
done
clear
View Answer play_arrow
In a eukaryotic cell splicing, capping and toiling steps
A)
are template dependent
done
clear
B)
occur in Cytoplasm
done
clear
C)
occur in nucleus
done
clear
D)
arc performed by SnRNPs
done
clear
View Answer play_arrow
Mark the incorrect match (with respect to pedigree analysis)
A)
Sex unspecified
done
clear
B)
Aflected individual
done
clear
C)
Consanguineous marriage resulting in affected son
done
clear
D)
Abortion
done
clear
View Answer play_arrow
Neurilemma is the name for
A)
Plasma membrane of axon
done
clear
B)
Coating formed by myelin sheath
done
clear
C)
Covering formed by schwann cells
done
clear
D)
Connective tissue covering of nerve fasciculus
done
clear
View Answer play_arrow
Cholecystokinin is the secretion of
A)
Duodenum that causes contraction of gall bladder
done
clear
B)
Goblet cells ofilcum, stimulates secretion of succus entericus
done
clear
C)
Liver and controls bile synthesis
done
clear
D)
Stomach that stimulates pancreas to release pancreatic juices
done
clear
View Answer play_arrow
Curdling of milk in small intestine would occur with the help of
A)
Rennin
done
clear
B)
Erepsin
done
clear
C)
Chymotrypsin
done
clear
D)
Trypsin
done
clear
View Answer play_arrow
Which structure in not related with respiration in frog?
A)
Lungs
done
clear
B)
Skin
done
clear
C)
Buccal cavity
done
clear
D)
Diaphragm
done
clear
View Answer play_arrow
Which of the following statements correctly defines Bohr effect?
A)
Rise in \[{{P}_{50}}\] with a decrease in \[C{{O}_{2}}\]concentration
done
clear
B)
Rise in \[{{P}_{50}}\] with a decrease in pH
done
clear
C)
Rise in \[{{P}_{50}}\] with a decrease in DPG concentration
done
clear
D)
Fall in \[{{P}_{50}}\] with a decrease in pH
done
clear
View Answer play_arrow
Pulse pressure is
A)
Systolic pressure
done
clear
B)
Diastolic pressure
done
clear
C)
Systolic pressure plus diastolic pressure
done
clear
D)
Difference between systolic and diastolic pressure
done
clear
View Answer play_arrow
Renal Portal System is very well developed in
A)
Vertebrates
done
clear
B)
Tetrapods
done
clear
C)
Anamniotes
done
clear
D)
Amniotes
done
clear
View Answer play_arrow
Ornithine cycle removes two waste products from blood in live, i.e.,
A)
Urea and Carbon dioxide
done
clear
B)
Carbon dioxide and ammonia
done
clear
C)
Ammonia and uric acid
done
clear
D)
Ammonia and urea
done
clear
View Answer play_arrow
Haeckel's biogenetic law received support from
A)
Serological studies
done
clear
B)
Morphological studies
done
clear
C)
Pa leontological studies
done
clear
D)
Embryological studies
done
clear
View Answer play_arrow
Sertoli cells perform the function of
A)
Maturation of sperm
done
clear
B)
Storage of sperms
done
clear
C)
Nourishment to sperms
done
clear
D)
Formation of testostcrone
done
clear
View Answer play_arrow
Factor that will not produce any change in Hardy Weinberg equilibrium is
A)
Directional selection
done
clear
B)
Stabilizing selection
done
clear
C)
Balanced poly morphism
done
clear
D)
Disruptive selection/random mating
done
clear
View Answer play_arrow
Releasing of second polar body from egg nucleus occurs
A)
Before entry of sperm
done
clear
B)
After finishing the fertilization
done
clear
C)
Due to attachment of sperm membrane with ovum
done
clear
D)
After entry of sperm nucleus but before completion of fertilization
done
clear
View Answer play_arrow
In an increased industrial polluted areas, some dark coloured peppered moth appear among the light coloured one. It is an example of
A)
Genetic drift
done
clear
B)
Mutation
done
clear
C)
Selection "Direction based
done
clear
D)
Random mating
done
clear
View Answer play_arrow
A thyroid hormone that is net stored as colloidal material in the thyroid follicles
A)
\[{{T}_{3}}\]
done
clear
B)
Thyroxine
done
clear
C)
Calcitonin
done
clear
D)
Triidothyronine
done
clear
View Answer play_arrow
When a person is suffering from poor renal re-absorption, which of these will not help in maintenance of blood volume?
A)
Reduced arterial pressure in kidneys
done
clear
B)
Reduced glomerular filtration
done
clear
C)
Increased ADH secretion
done
clear
D)
Increased arterial pressure in kidneys
done
clear
View Answer play_arrow
A person passes out excessive urine and drinks much whaler but his glucose level is normal. It is due to
A)
decrease in ADH secretion
done
clear
B)
decrease in insulin secretion
done
clear
C)
increased secretion of glucagon
done
clear
D)
fall in glucose concentration released in urine
done
clear
View Answer play_arrow
Craniosacral outflow is
A)
Parasympathetic nervous system and it is cholinergic
done
clear
B)
Sympathetic nervous system and it is cholinergic
done
clear
C)
Sympathetic nervous system and it is adrenergic
done
clear
D)
Parasympathetic nervous system and it is adrenergic
done
clear
View Answer play_arrow
Kingdom fungi is classified into various classes on the basis of all, except
A)
Morph ology of mycelium
done
clear
B)
Mode of spore formation
done
clear
C)
Types of fruiting bodies
done
clear
D)
Method of plasmogamy
done
clear
View Answer play_arrow
If the margins of sepals or petals overlap one another but not in any particular direction like Cassia, the acstivation is
A)
Vexillary
done
clear
B)
Twisted
done
clear
C)
Imbricate
done
clear
D)
Valvate
done
clear
View Answer play_arrow
Choose odd one out with respect to facilitated diffusion
A)
Highly selective
done
clear
B)
Uphill and needs ATP
done
clear
C)
Transport achieves saturation
done
clear
D)
Affected by protein inhibitors
done
clear
View Answer play_arrow
Plant hormone that can be used to improve the shape of apples and to increase length of grape stalks is
A)
Cytokinins
done
clear
B)
Auxins
done
clear
C)
Gibberellins
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
Choose correct option with respect to adapter molecule
A)
It has first loop for enzyme binding from 3' end
done
clear
B)
There are no tRNAs for stop codons
done
clear
C)
Previously called as sRNA soluble in the cell
done
clear
D)
The primary structure of tRNA looks like a clover leaf
done
clear
View Answer play_arrow
Promoter is a transcription unit is located at
A)
3' end upstream of coding strand
done
clear
B)
5' end downstream of a non-template strand
done
clear
C)
5'end upstream of coding strand
done
clear
D)
5' end downstream non-coding strand
done
clear
View Answer play_arrow
Bioactive molecule used as an immunosuppressive agent in organ transplant is produced by
A)
Tolypocladuminflatum
done
clear
B)
Monascus purpureus
done
clear
C)
Candida lipolytica
done
clear
D)
Trichodcrma polysporum
done
clear
View Answer play_arrow
Pyramid of number in tree ecosystem with predatory food chain is
A)
Triangular
done
clear
B)
Upright
done
clear
C)
Spindle shaped
done
clear
D)
Inverted
done
clear
View Answer play_arrow
Particulate matters in air can be removed most efficiently by the use of
A)
Catalytic converters in exhaust of automobiles
done
clear
B)
Scrubbers
done
clear
C)
Electrostatic precipitators
done
clear
D)
Euro-III emission norms
done
clear
View Answer play_arrow

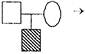 Consanguineous marriage resulting in affected son
Consanguineous marriage resulting in affected son 