A) 7 cm
B) 5 cm
C) 4.5 cm
D) 2.3 cm
Correct Answer: B
Solution :
Key Idea: An isothermal process obeys Boyles law. Since process is isothermal the total pressure of air inside the bubble is same as excess of pressure given by \[=\frac{3}{2}\times 0.01\times 100\]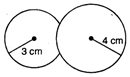 where T is surface tension and R is radius . Also an isothermal process obeys Boyles law hence, PV= constant. Let R be the radius of coalesce system then \[=1.5%\] \[Y=3K(1-2\sigma )\]\[{{I}_{1}}\] \[{{I}_{2}}\] Given, \[\phi \] and \[I={{I}_{1}}+{{I}_{2}}+2\sqrt{{{I}_{1}}{{I}_{2}}}\cos \phi \] \[\cos \phi =-1\] \[\phi =\pi ,3\pi ,5\pi ,.................\] \[\phi =(2\pi +1)\pi \,n=1,2,3,..............\]
where T is surface tension and R is radius . Also an isothermal process obeys Boyles law hence, PV= constant. Let R be the radius of coalesce system then \[=1.5%\] \[Y=3K(1-2\sigma )\]\[{{I}_{1}}\] \[{{I}_{2}}\] Given, \[\phi \] and \[I={{I}_{1}}+{{I}_{2}}+2\sqrt{{{I}_{1}}{{I}_{2}}}\cos \phi \] \[\cos \phi =-1\] \[\phi =\pi ,3\pi ,5\pi ,.................\] \[\phi =(2\pi +1)\pi \,n=1,2,3,..............\]
You need to login to perform this action.
You will be redirected in
3 sec
