A) Tartaric acid, propanoic acid
B) Succinic acid, succinic anhydride
C) Maleic anhydride, maleic acid
D) Methyl malonic acid, propanoic acid
Correct Answer: D
Solution :
\[\underset{A}{\mathop{{{C}_{4}}{{H}_{6}}{{O}_{4}}}}\,\xrightarrow{\Delta }\underset{B}{\mathop{{{C}_{3}}{{H}_{6}}{{O}_{2}}}}\,\] From above equation, it is clear that \[C{{O}_{2}}\] is lost due to heating. Thus, A is a dibasic acid with two -COOH groups on same carbon atom (gem position). Thus, A is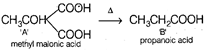 \[\xrightarrow[\Delta ]{Soda\text{ }lime}\underset{\begin{smallmatrix} C \\ ethane \end{smallmatrix}}{\mathop{C{{H}_{3}}C{{H}_{3}}}}\,\] For 1 mole of C again, \[C{{H}_{3}}C{{H}_{3}}(mov\,\,wt=30)\] \[\therefore \] 30 g of C occupies volume = 22.42 \[\therefore \] 1 g of C with occupy volume \[=\frac{22.4}{30}\] \[\simeq 0.75\text{ }L\]
\[\xrightarrow[\Delta ]{Soda\text{ }lime}\underset{\begin{smallmatrix} C \\ ethane \end{smallmatrix}}{\mathop{C{{H}_{3}}C{{H}_{3}}}}\,\] For 1 mole of C again, \[C{{H}_{3}}C{{H}_{3}}(mov\,\,wt=30)\] \[\therefore \] 30 g of C occupies volume = 22.42 \[\therefore \] 1 g of C with occupy volume \[=\frac{22.4}{30}\] \[\simeq 0.75\text{ }L\]
You need to login to perform this action.
You will be redirected in
3 sec
