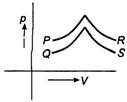
A) P
B) Q
C) R
D) S
Correct Answer: D
Solution :
For p-V diagrams P and Q pressure increases with increase in volume. This is possible when the temperature of the gas increases. Hence, exchange of heat takes place. For p-V diagrams, R and 5 pressure decreases with increase in volume but the slope of curve is larger for S. Therefore, it is for an adiabatic process for which no exchange of heat takes place.You need to login to perform this action.
You will be redirected in
3 sec
