A) cone. \[{{H}_{2}}S{{O}_{4}}\]
B) sodium alkoxide
C) dry silver oxide
D) Grignard reagent
Correct Answer: D
Solution :
\[\underset{lower\,ether}{\mathop{C{{H}_{3}}OC{{H}_{3}}}}\,\xrightarrow{C{{l}_{2}}/hv}\underset{\begin{smallmatrix} \text{ }\!\!\alpha\!\!\text{ -chlorodimethyl}\,\text{ether} \\ \text{(lower}\,\text{halogenated}\,\text{ether)} \end{smallmatrix}}{\mathop{C{{H}_{3}}OC{{H}_{2}}Cl}}\,\] \[C{{H}_{3}}OC{{H}_{2}}Cl+\underset{\begin{smallmatrix} Grignard \\ reagent \end{smallmatrix}}{\mathop{C{{H}_{3}}MgBr}}\,\xrightarrow[{}]{Dry\,etger}\]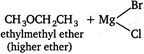
You need to login to perform this action.
You will be redirected in
3 sec
