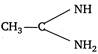
 (2) \[C{{H}_{3}}-C{{H}_{2}}-N{{H}_{2}}\] (3) \[{{(C{{H}_{3}})}_{2}}NH\] (4) \[C{{H}_{3}}-\overset{\begin{smallmatrix} O \\ |\,\,| \end{smallmatrix}}{\mathop{C}}\,-N{{H}_{2}}\]
(2) \[C{{H}_{3}}-C{{H}_{2}}-N{{H}_{2}}\] (3) \[{{(C{{H}_{3}})}_{2}}NH\] (4) \[C{{H}_{3}}-\overset{\begin{smallmatrix} O \\ |\,\,| \end{smallmatrix}}{\mathop{C}}\,-N{{H}_{2}}\]
A) \[2>1>3>4\]
B) \[1>3>2>4\]
C) \[3>1>2>4\]
D) \[1>2>3>4\]
Correct Answer: B
Solution :
Protonated compound of I is stabilised by two equivalent resonating structures, hence compound I should accept\[{{H}^{+}}\]most readily. \[{{H}_{3}}C-\overset{\begin{smallmatrix} O \\ |\,| \end{smallmatrix}}{\mathop{C}}\,-N{{H}_{2}}\xrightarrow[{}]{{{H}^{+}}}{{H}_{3}}C-\overset{\begin{smallmatrix} N{{H}_{2}} \\ |\,| \end{smallmatrix}}{\mathop{C}}\,-N{{H}_{2}}\underset{{}}{\overset{{}}{\longleftrightarrow}}\] \[{{H}_{3}}C-\overset{\begin{smallmatrix} ^{+}N{{H}_{2}} \\ |\,\,| \end{smallmatrix}}{\mathop{C}}\,-N{{H}_{2}}\underset{{}}{\overset{{}}{\longleftrightarrow}}{{H}_{3}}C-\overset{\begin{smallmatrix} N{{H}_{2}} \\ |\,| \end{smallmatrix}}{\mathop{C}}\,=N{{H}_{2}}\] Further\[2{}^\circ \]amines are more basic than\[1{}^\circ \]amine white amides are least basic because electron pair on nitrogen is involved in delocalisation \[{{H}_{3}}-\overset{\begin{smallmatrix} \bullet \bullet \\ {}_{\bullet }^{\bullet }O_{\bullet }^{\bullet } \\ || \end{smallmatrix}}{\mathop{C}}\,-\overset{\bullet \,\bullet }{\mathop{N}}\,{{H}_{2}}\underset{{}}{\overset{{}}{\longleftrightarrow}}{{H}_{3}}C-\overset{\begin{smallmatrix} \bullet \bullet \\ {}_{\bullet }^{\bullet }O_{\bullet }^{\bullet } \\ || \end{smallmatrix}}{\mathop{C}}\,-N{{\overset{+}{\mathop{H}}\,}_{2}}\] So, the correct order of basicity is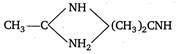 \[>C{{H}_{3}}-C{{H}_{2}}-N{{H}_{2}}>C{{H}_{3}}{{-}^{*}}\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,N{{H}_{2}}\]
\[>C{{H}_{3}}-C{{H}_{2}}-N{{H}_{2}}>C{{H}_{3}}{{-}^{*}}\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,N{{H}_{2}}\]
You need to login to perform this action.
You will be redirected in
3 sec
