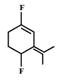
A)
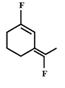
B)
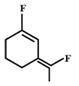
C)
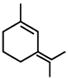
D)
Correct Answer: C
Solution :
Among the substituent attached to the given compounds, Fluorine has maximum polarity. So it will push dipole moment towards itself. In compound in option A the two F groups are attached opposite to each other thus net dipole moment will cancel each other and reduce its polarity. In compound in option B the F groups are attached in slightly opposite direction thus this also decreases its polarity. But the compound in option C has the two F groups along same direction. Thus net dipole moment will increase in this direction thus it will have maximum polarity. Thus the compound in option C has maximum polarity.You need to login to perform this action.
You will be redirected in
3 sec
