A) Act as both oxidising and reducing agent
B) Two OH. bond lie in the same plane
C) Pale blue liquid
D) Can be oxidised by ozone
Correct Answer: B
Solution :
\[{{2}^{3n}}\] is a pale blue liquid, it can be oxidised by ozone. \[{{2}^{2n}}\] acts as both oxidising and reducing agent. The value of dipole moment of \[{{2}^{n}}\] is 2.1 D. Which suggest it cannot be planar in fact it has open book like structure.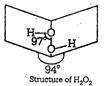 Two O ? H lie in different plane
Two O ? H lie in different plane
You need to login to perform this action.
You will be redirected in
3 sec
