A) Glucose
B) Water
C) Both and
D) None of these
Correct Answer: B
Solution :
The freezing point of a, liquid is that temperature at which the liquid and its solid state exist in equilibrium with each other and have same vapour pressure. Due to lower vapour pressure of the solution, solid form of solution separates out a lower temperature. The decrease in freezing point is called depression in freezing point. Thus if an aqueous solution of glucose is allowed to freeze then crystal of water will be separated out first.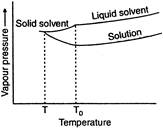
You need to login to perform this action.
You will be redirected in
3 sec
