A)
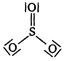
B)
![]()
C)
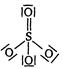
D)
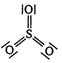
Correct Answer: D
Solution :
| Formal charges help in the selection of the lowest energy structure from a number of possible Lewis structures for a given species. |
| Generally the lowest energy structure is the one with the smallest formal charges on the atoms. |
| Formal charge on an atom: |
| = total no. of valence electrons-non-bonding electrons \[-\frac{1}{2}\times \] bonding electrons. |
| For Lewis structure of \[S{{O}_{3}}\] |
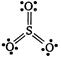 |
| Formal charge on S atom \[=6-0-\frac{1}{2}\times 12=0\] |
| Formal charge on three O atoms |
| \[=6-4-\frac{1}{2}\times 4=0\] |
You need to login to perform this action.
You will be redirected in
3 sec
