A) Ethanal
B) Propanal
C) Butanone
D) Propanone
Correct Answer: A
Solution :
As we move from ethanal to propanal, to propanone to butanone, the \[+I\] inductive effect of alkyl group increases, as a result, the \[+ve\] charge on the carbon atom of the carbonyl group progressively decreases and hence, attack by nucleophile becomes slower and slower.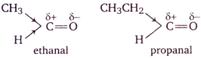
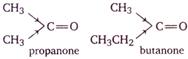 Thus, the reactivity increases in the order: butanone < propanone < propanal < ethanol
Thus, the reactivity increases in the order: butanone < propanone < propanal < ethanol
You need to login to perform this action.
You will be redirected in
3 sec
