A) \[HCl\]
B) \[C{{l}_{2}}\]
C) \[PC{{l}_{5}}\]
D) aq \[NaHS{{O}_{3}}\]
Correct Answer: A
Solution :
\[HCl\]is a strong inorganic acid, hence it does not react with ethanal whereas with other given reagents, it reacts in the following manner: \[\underset{ethanal}{\mathop{C{{H}_{3}}CHO}}\,+C{{l}_{2}}\xrightarrow[{}]{{}}\underset{chloral}{\mathop{CC{{l}_{3}}CHO}}\,\] \[C{{H}_{3}}CHO+PC{{l}_{5}}\xrightarrow[{}]{{}}C{{H}_{3}}CHC{{l}_{2}}\]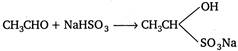 \[C{{H}_{3}}CHO+HCl\xrightarrow[{}]{{}}No\text{ }reaction.\]
\[C{{H}_{3}}CHO+HCl\xrightarrow[{}]{{}}No\text{ }reaction.\]
You need to login to perform this action.
You will be redirected in
3 sec
