A) formic acid > acetic acid > propionic acid
B) cyclohexanol < phenol < benzoic acid
C) benzamide < aniline < cyclohexylamine
D) \[FC{{H}_{2}}COOH>ClC{{H}_{2}}COOH>BrC{{H}_{2}}COOOH\]
Correct Answer: C
Solution :
Usually presence of electron withdrawing groups such as\[F,Cl,\]CHO etc increases the acidic strength and presence of electron releasing groups such as\[-R,-OC{{H}_{3}}\]etc decreases the acidic strength. Aromatic compounds are more acidic than their corresponding aliphatic compounds due to resonance stabilisation. Hence, the correct order of acidic strength is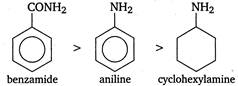
You need to login to perform this action.
You will be redirected in
3 sec
